Cardiovascular Clinical Trials Market Size Outlook and Demand Growth Across 2026–2030
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Market Value Is Anticipated For The Cardiovascular Clinical Trials Market Over The Period 2026–2030?
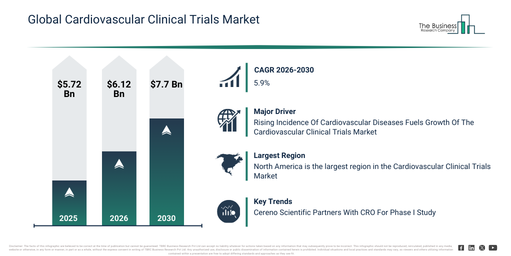
The cardiovascular clinical trials market size has experienced significant expansion in recent years. It is anticipated to increase from $5.72 billion in 2025 to $6.12 billion in 2026, achieving a compound annual growth rate (CAGR) of 7.0%. This historical growth can be primarily ascribed to the rising incidence of cardiovascular diseases, the broadening scope of global clinical research endeavors, a growing number of regulatory approvals for innovative therapies, the existence of well-established trial infrastructure, and the proliferation of multicenter studies.
The cardiovascular clinical trials market is anticipated to expand significantly over the next few years, projected to reach $7.7 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 5.9%. This growth during the forecast period can be attributed to several factors, including an escalating demand for more rapid trial execution, the increasing adoption of AI-driven trial analytics, the broader implementation of remote patient engagement strategies, a heightened emphasis on patient-centric trial designs, and greater investment in cardiovascular drug pipelines. Key trends expected during this period involve the rising uptake of decentralized trial models, an increased utilization of digital patient monitoring tools, the progressive integration of biomarkers into study designs, an expansion in the generation of real-world evidence, and an augmented focus on adaptive trial designs.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=15532&type=smp
What Drivers Are Expected To Influence The Cardiovascular Clinical Trials Market During The Forecast Period?
The increasing occurrence of cardiovascular conditions is projected to fuel the expansion of the cardiovascular clinical trials market in the future. These diseases encompass various ailments impacting the heart and blood vessels, such as coronary artery disease, heart failure, and stroke. The rise in cardiovascular disease cases is linked to several factors, including inactive lifestyles, poor dietary habits, tobacco use, obesity, hypertension, diabetes, and inherited susceptibilities. Clinical trials in the cardiovascular field are instrumental in finding and confirming novel medications to better manage risk factors like hypertension, elevated cholesterol, and diabetes, all of which are major contributors to CVDs. For example, data released in September 2024 by the Minnesota Department of Health, a US-based state health agency, indicated that about 30% of adults in Minnesota reported high blood pressure in 2023, affecting close to 1.4 million individuals. Furthermore, in 2022, conditions related to hypertension were identified as the primary or contributing factor in the deaths of 14,225 residents, accounting for nearly 28% of all fatalities statewide. Consequently, the increasing prevalence of cardiovascular diseases is propelling the expansion of the cardiovascular clinical trials market.
How Is The Cardiovascular Clinical Trials Market Divided Into Its Major Segments?
The cardiovascular clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV
2) By Study Design: Interventional, Observational, Expanded Access
3) By Indication: Acute Coronary Syndrome, Coronary Artery Disease, Ischemic Heart Disease, Pulmonary Arterial Hypertension, Stroke, Cardiac Arrhythmias, Heart Failure, Other Indications
Subsegments:
1) By Phase I: First-in-Human Trials, Dose Escalation Studies, Safety and Tolerability Studies, Pharmacokinetics And Pharmacodynamics Studies
2) By Phase II: Efficacy Studies, Optimal Dosage And Administration Route Studies, Early Safety And Efficacy Trials, Biomarker Development Trials
3) By Phase III: Large-Scale Efficacy Trials, Randomized Controlled Trials (RCTs), Long-Term Safety And Efficacy Studies, Multicenter Trials
4) By Phase IV: Post-Marketing Surveillance, Long-Term Safety Studies, Real-World Evidence (RWE) Studies, Comparative Effectiveness Research
What Major Market Trends Are Driving Changes In The Cardiovascular Clinical Trials Market?
Leading firms involved in the cardiovascular clinical trials market are concentrating on forming strategic alliances, including those with contract research organizations (CROs), to broaden their distribution reach and access a larger client base. Such CRO collaborations are vital within the clinical research sector, entailing cooperation among diverse entities to deliver specialized assistance and services for different facets of clinical trials and studies. An example of this is the September 2023 collaboration between Cereno Scientific AB, a biopharmaceutical company from Sweden, and Clinical Trial Consultants (CTC), a full-service CRO based in Sweden specializing in clinical conduct. Their partnership aimed to execute a Phase I study for CS014, a histone deacetylase inhibitor designed to prevent arterial and venous thrombosis. CTC will additionally provide support for Phase I preparatory steps, including the formulation of the study protocol and the clinical trial application process for this study, which is slated to take place in Sweden. This initial human Phase I trial is slated to commence during the first half of 2024. This joint effort represents a considerable stride forward in enhancing cardiovascular health research.
Who Are The Prominent Organizations Shaping The Cardiovascular Clinical Trials Market?
Major companies operating in the cardiovascular clinical trials market are Pfizer Inc., Johnson & Johnson, AstraZeneca PLC, Novartis AG, Eli Lilly and Company, Amgen Inc., Boehringer Ingelheim International GmbH, Merck & Co Inc., Sanofi, Baxter International Inc., IQVIA Holdings Inc., ICON plc, Syneos Health Inc., PPD Inc., Medpace Holdings Inc., Worldwide Clinical Trials, SGS SA, Charles River Laboratories International Inc., WuXi AppTec Co Ltd, Gilead Sciences Inc.
Read the full cardiovascular clinical trials market report here:
Which Region Is Forecast To Lead The Cardiovascular Clinical Trials Market In Terms Of Market Size?
North America was the largest region in the cardiovascular clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the cardiovascular clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Cardiovascular Clinical Trials Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=15532&type=smp
Browse Through More Reports Similar to the Global Cardiovascular Clinical Trials Market 2026, By The Business Research Company
Hypertrophic Cardiomyopathy Treatment Global Market Report
Heart Attack Diagnostic Global Market Report
https://www.thebusinessresearchcompany.com/report/heart-attack-diagnostic-global-market-report
Cardiovascular Drugs Global Market Report
https://www.thebusinessresearchcompany.com/report/cardiovascular-drugs-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model