Cardiovascular Clinical Trials Market Forecast: How Big Will It Be by 2030?
Uncover key drivers, emerging technologies, and competitive movements shaping the cardiovascular clinical trials market from 2026–2035 with trusted insights from The Business Research Company
What size range is anticipated for the Cardiovascular Clinical Trials Market from 2026 to 2030?
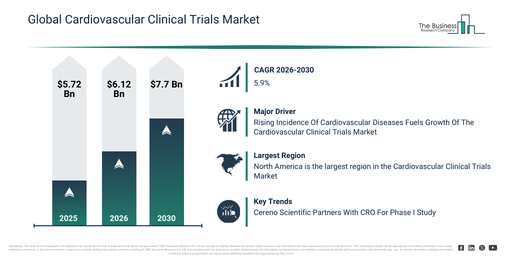
The cardiovascular clinical trials market size has seen significant growth recently. It is anticipated to expand from $5.72 billion in 2025 to $6.12 billion in 2026, achieving a compound annual growth rate (CAGR) of 7.0%. This historical growth can be linked to the increasing burden of cardiovascular diseases, the expansion of global clinical research initiatives, a rise in regulatory approvals for new therapies, the availability of existing trial infrastructure, and the increase in multicenter studies.
The cardiovascular clinical trials market is poised for robust expansion in the upcoming years, with its size anticipated to reach $7.7 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 5.9%. This projected growth can be ascribed to several factors, including an escalating demand for faster trial execution, the increasing deployment of AI-driven trial analytics, wider implementation of remote patient engagement strategies, a heightened focus on patient-centric trial designs, and greater investment in cardiovascular drug pipelines. Prominent trends throughout this period encompass the rising adoption of decentralized trial models, an increased utilization of digital patient monitoring tools, deeper integration of biomarkers into study designs, an expansion in real-world evidence generation, and a sharpened emphasis on adaptive trial designs.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=15532&type=smp
What Drivers Are Affecting Demand In The Cardiovascular Clinical Trials Market?
The rising occurrence of cardiovascular ailments is projected to fuel the expansion of the cardiovascular clinical trials market in the future. Cardiovascular disease encompasses a range of conditions impacting the heart and blood vessels, such as coronary artery disease, heart failure, and stroke. A heightened prevalence of cardiovascular diseases is attributed to multiple elements, including inactive lifestyles, poor dietary habits, tobacco use, excess weight, elevated blood pressure, diabetes, and inherited predispositions. Cardiovascular clinical trials play a crucial role in identifying and confirming novel medications capable of more effectively managing risk factors like hypertension, high cholesterol, and diabetes, which are major contributors to CVDs. For example, in September 2024, as per information released by the Minnesota Department of Health, a US-based state health agency, roughly 30% of adults in Minnesota stated they had high blood pressure in 2023, amounting to nearly 1.4 million individuals, and in 2022, illnesses related to hypertension were the primary or secondary cause of death for 14,225 residents, constituting almost 28% of all fatalities in the state. Consequently, the increasing prevalence of cardiovascular diseases is propelling the growth of the cardiovascular clinical trials market.
Which Segment Groups Are Influencing The Cardiovascular Clinical Trials Market?
The cardiovascular clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV
2) By Study Design: Interventional, Observational, Expanded Access
3) By Indication: Acute Coronary Syndrome, Coronary Artery Disease, Ischemic Heart Disease, Pulmonary Arterial Hypertension, Stroke, Cardiac Arrhythmias, Heart Failure, Other Indications
Subsegments:
1) By Phase I: First-in-Human Trials, Dose Escalation Studies, Safety and Tolerability Studies, Pharmacokinetics And Pharmacodynamics Studies
2) By Phase II: Efficacy Studies, Optimal Dosage And Administration Route Studies, Early Safety And Efficacy Trials, Biomarker Development Trials
3) By Phase III: Large-Scale Efficacy Trials, Randomized Controlled Trials (RCTs), Long-Term Safety And Efficacy Studies, Multicenter Trials
4) By Phase IV: Post-Marketing Surveillance, Long-Term Safety Studies, Real-World Evidence (RWE) Studies, Comparative Effectiveness Research
What Trends Are Reshaping The Dynamics Of The Cardiovascular Clinical Trials Market?
Major companies operating in the cardiovascular clinical trials market are prioritizing the formation of strategic alliances, such as partnerships with contract research organizations (CROs), to expand their distribution channels and reach a broader customer base. A CRO partnership serves a critical function in the clinical research industry, entailing collaborations among diverse companies to deliver specialized services and assistance for various components of clinical trials and research. For example, in September 2023, Cereno Scientific AB, a biopharmaceutical company from Sweden, joined forces with Clinical Trial Consultants (CTC), a Sweden-based full-service CRO focused on clinical conduct, to execute a Phase I study for CS014, a histone deacetylase inhibitor aimed at averting arterial and venous thrombosis. CTC will also contribute to Phase I preparation processes like the development of study protocols and the clinical trial application procedure for the study, which will be conducted in Sweden. This inaugural first-in-human Phase I trial is projected to commence in the first half of 2024. This collaboration represents a substantial step forward in advancing research into cardiovascular health.
Who Are The Prominent Global Companies Shaping The Cardiovascular Clinical Trials Market Landscape?
Major companies operating in the cardiovascular clinical trials market are Pfizer Inc., Johnson & Johnson, AstraZeneca PLC, Novartis AG, Eli Lilly and Company, Amgen Inc., Boehringer Ingelheim International GmbH, Merck & Co Inc., Sanofi, Baxter International Inc., IQVIA Holdings Inc., ICON plc, Syneos Health Inc., PPD Inc., Medpace Holdings Inc., Worldwide Clinical Trials, SGS SA, Charles River Laboratories International Inc., WuXi AppTec Co Ltd, Gilead Sciences Inc.
Access The Complete Report For Deeper Market Insights:
Which Geographic Areas Are Emerging As Strong Markets For The Cardiovascular Clinical Trials Market?
North America was the largest region in the cardiovascular clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the cardiovascular clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Cardiovascular Clinical Trials Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=15532&type=smp
Browse Through More Reports Similar to the Global Cardiovascular Clinical Trials Market 2026, By The Business Research Company
Cardiovascular Digital Solutions Market Report 2026
Cardiovascular Drugs Market Report 2026
https://www.thebusinessresearchcompany.com/report/cardiovascular-drugs-global-market-report
Cardiovascular Health Supplements Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model