Clinical Trial Supplies Market Outlook 2025: Mapping Growth, Innovation, and Regional Shifts
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
What Is the Current and Projected Market Size of the Clinical Trial Supplies Market Through 2034?
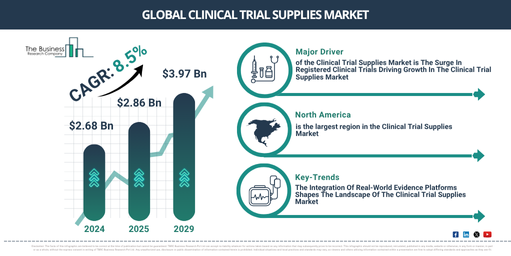
The market size for clinical trial supplies has seen substantial growth in the past few years. It’s projected to escalate from $2.68 billion in 2024 to $2.86 billion in 2025, bearing a compound annual growth rate (CAGR) of 6.9%. The growth seen in the historical timeframe can be linked to the rise in chronic diseases, strict regulatory standards, increased need for clinical trials to innovate new treatments, and the growing complexity of clinical trials.
In the coming few years, the market size of clinical trial supplies is anticipated to experience substantial growth, with projections indicating it will reach $3.97 billion by 2029, following a compound annual rate (CAGR) of 8.5%. The predicted rise during this forecast duration can be linked to factors such as the globalization of clinical trials, the growth of cell and gene therapy trials, a heightened emphasis on patient-focused trials, an increase in the number of biologics and biosimilar drugs undergoing trials, and the emergence of virtual and decentralized trials. Key trends during the forecast period include the incorporation of artificial intelligence (AI) and predictive analytics, 3D printing, an increasing emphasis on sustainability and eco-friendliness in clinical trial supply management, surging R&D investments by biopharmaceutical and pharmaceutical companies, and progress in blockchain technology.
Download a free sample to assess the report’s scope and structure:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12133&type=smp
What External and Internal Drivers Are Contributing to the Growth of theClinical Trial Supplies Market?
The clinical trial supplies market is predicted to expand due to the rising number of registered clinical trials. Clinical trials are essential research studies involving human subjects that assess the safety and efficacy of new healthcare treatments, procedures, or diagnostic methods. Key elements for accomplishing successful clinical trials include clinical trial supplies, such as pharmaceuticals, medical devices, and other vital materials. These components facilitate the research progression, guarantee precise data acquisition, and safeguard patients’ wellbeing. For clarification, ClinicalTrials.gov, a branch of the National Institutes of Health (NIH), an American government agency dedicated to overseeing and funding medical research, reported approximately 437,533 registered clinical trials in May 2023. This figure shows an increase from the 399,499 studies registered in 2022, spanning all 50 states of the United States and 221 different countries. Consequently, the surge in registered clinical trials is fuelling the growth of the clinical trial supplies market.
What Segment Types Define the Clinical Trial Supplies Market Structure?
The clinical trial suppliesmarket covered in this report is segmented –
1) By Services: Logistics And Distribution, Storage And Retention, Supply Chain Management, Packaging, Labeling, And Blinding, Manufacturing, Comparator Sourcing
2) By Clinical Phases: Phase I, Phase II, Phase III, Phase IV
3) By Therapeutic Use: Oncology, Central Nervous System (CNS), Cardiovascular, Infectious Disease, Metabolic Disorders, Other Therapeutic Uses
4) By End User: Pharmaceutical And Biotech companies, Contract Research Organization (CRO), Medical Device Companies
Subsegments:
1) By Logistics And Distribution: Transportation, Inventory Management, Temperature-Controlled Shipping, Customs Clearance
2) By Storage And Retention: Short-term Storage, Long-term Storage, Controlled Environment Storage
3) By Supply Chain Management: Planning And Forecasting, Supplier Management, Risk Management, Compliance Management
4) By Packaging, Labeling, And Blinding: Primary Packaging, Secondary Packaging, Label Design And Printing, Blinding Techniques
5) By Manufacturing: Active Pharmaceutical Ingredient (API) Production, Formulation Development, Batch Production
6) By Comparator Sourcing: Sourcing Of Reference Products, Quality Control And Assurance, Regulatory Compliance For Comparators
Request customized data on this market:
https://www.thebusinessresearchcompany.com/customise?id=12133&type=smp
Which Geographic Areas Hold the Strongest Growth Potential in the Clinical Trial Supplies Market?
North America was the largest region in the clinical trial supplies market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the clinical trial supplies market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
What Long-Term Trends Are Transforming the Competitive Landscape of the Clinical Trial Supplies Market?
The emergence of sophisticated real-world evidence platforms is a crucial trend that is gaining traction within the clinical trial supplies market. By adopting cutting-edge technology, key players in this market are revolutionizing how healthcare decisions, drug development, and patient care are approached, largely through the use of real-world data to develop evidence-backed perspectives. This has been vital in maintaining a strong market position. Take the case of Komodo Health in April 2023, an American healthcare tech firm specializing in patient recruitment, site selection, and real-time monitoring of data for clinical trials. The company introduced MapEnhance, which is part of an integrated real-world evidence (RWE) tech platform with a network of specialty data partners. MapEnhance provides customers an in-depth understanding of clinical events and patient demographics, delivering unparalleled enlightenment. The MapEnhance feature has a wide range of capabilities. These include precision molecular diagnostics across diverse therapy areas, high-yield regular laboratory diagnostics, enabling earlier detection of diseases, and electronic medical records that encapsulate clinical interactions. Added bonuses are inpatient and outpatient facility price lists that provide detailed cost analysis and usage data across varied hospital environments.
View the full report here:
https://www.thebusinessresearchcompany.com/report/clinical-trial-supplies-global-market-report
What Is the Definition of the Clinical Trial Supplies Market?
Clinical trial supplies refer to the materials, products, and equipment used during clinical trials. Clinical trial supplies play a crucial role in the successful execution of clinical trials by providing the necessary resources for testing and evaluation.
Purchase the full report and get a swift delivery:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=12133
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model