Growth In The Companion Diagnostics Market Is Being Driven By The Surge In Targeted Therapies Fueling The Expansion Of Companion Diagnostics
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
How Is The Market Size Of The Companion Diagnostics Market Expected To Change From 2026 To 2030?
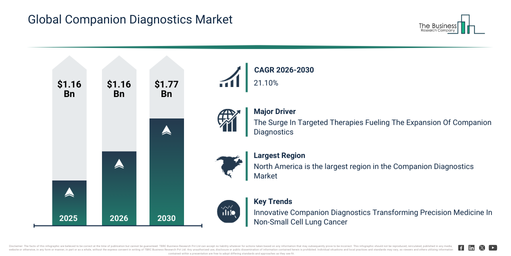
The companion diagnostics market size has shown substantial expansion in recent years. It is anticipated to increase from $8.69 billion in 2025 to $10.54 billion in 2026, registering a compound annual growth rate (CAGR) of 21.3%. This historical growth can be linked to the rise in targeted cancer therapies, the widening scope of oncology clinical trials, stronger collaborations between pharmaceutical and diagnostics companies, an increase in cancer incidence rates, and regulatory approvals for companion tests.
The companion diagnostics market is projected to experience substantial expansion over the coming years. This market is anticipated to reach a valuation of $22.7 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 21.1%. Several factors are driving this anticipated growth, including a heightened demand for tailored treatment options, increased financial commitments to precision medicine, the broadening of immuno-oncology research and development, the increasing integration of AI in diagnostic analysis, and a stronger regulatory focus on the joint development of diagnostics and drugs. Key developments expected during this period involve a greater embrace of precision oncology diagnostics, the deeper embedding of companion diagnostics into pharmaceutical development processes, the expanding utilization of NGS-based diagnostic platforms, the proliferation of therapies guided by biomarkers, and an intensified commitment to personalized medicine approaches.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=7038&type=smp
Which Strong Drivers Are Impacting The Companion Diagnostics Market Growth?
The increasing prevalence of targeted therapies is anticipated to stimulate the expansion of the companion diagnostics market. Targeted therapy functions as a pharmacological treatment that aims at particular characteristics of cancer cells to prevent the disease from growing and spreading. These medications circulate throughout the body but exert a more precise effect than chemotherapy, frequently resulting in fewer adverse reactions. Companion diagnostic testing, based on biological markers for targeted cancer treatment, is becoming an indispensable component of personalized cancer care. For instance, in February 2024, the Personalized Medicine Coalition, a US-based hospital and health care organization, reported that in 2023, the FDA approved 16 new personalized treatments for rare disease patients, an increase from 6 in 2022. Therefore, the rise in targeted therapies significantly contributes to the growth of the companion diagnostics market.
What Segment Categories Shape The Companion Diagnostics Market Segment Landscape?
The companion diagnostics market covered in this report is segmented –
1) By Product And Service: Assays, Kits And Reagents, Software And Services
2) By Technology: Polymerase Chain Reaction, Next-Generation Sequencing, In Situ Hybridization, Immunohistochemistry
3) By Indication: Lung Cancer, Breast Cancer, Colorectal Cancer, Leukemia, Melanoma
4) By End-User: Pharmaceutical And Biopharmaceutical Companies, Reference Laboratories, Contract Research Organizations
Subsegments:
1) By Assays: Polymerase Chain Reaction (PCR) Assays, Immunohistochemistry (IHC) Assays, Next-Generation Sequencing (NGS) Assays, In Situ Hybridization (ISH) Assays
2) By Kits And Reagents: Diagnostic Kits, Detection Reagents, Control Reagents
3) By Software And Services: Data Analysis Software, Interpretation Software, Custom Development Services, Consulting and Support Services
Which Trends Are Contributing To Changes In The Companion Diagnostics Market?
Leading companies within the companion diagnostics market are forming partnerships to create advanced treatment solutions, aiming to improve personalized medicine, enhance patient results, and maximize the efficacy of targeted therapies. Such corporate alliances are expected to hasten the progress and market availability of companion diagnostics (CDx) for treatments in hematology and oncology. For example, in January 2024, Agilent Technologies, Inc., a US-based company supplying laboratory instruments, software, services, and consumables, announced an agreement with Incyte, a US-based pharmaceutical company. This collaboration will leverage Agilent’s specialized knowledge and extensive experience in developing companion diagnostics (CDx) to aid in the creation and commercialization of Incyte’s hematology and oncology product range. This partnership enables Agilent to enlarge its CDx portfolio by incorporating novel biomarkers, while Incyte benefits from Agilent’s proficiency in assay development, obtaining regulatory approvals, and commercialization efforts, all of which support clinical trials and CDx registration in the U.S. and Europe.
Who Are The Companies Operating Across The Companion Diagnostics Market Value Chain?
Major companies operating in the companion diagnostics market are F Hoffmann-La Roche Ltd., Agilent Technologies Inc., Qiagen NV, Thermo Fisher Scientific Inc., Abbott Laboratories Inc., Amoy Diagnostics Co. Ltd., Danaher Corporation, Guardant Health Inc., Illumina Inc., Myriad Genetics Inc., Siemens Healthcare GmbH, Almac Group, Caris Life Sciences, Cepheid Inc., Diaceutics plc, Epic Sciences Inc., Foundation Medicine Inc., Genomic Health Inc., Grail Inc., Hologic Inc., HTG Molecular Diagnostics Inc., Inivata Ltd., Invivoscribe Inc., MolecularMD Corporation, Natera Inc., NeoGenomics Laboratories Inc., Personal Genome Diagnostics Inc., Precision Therapeutics Inc., Prometheus Laboratories Inc., Sysmex Corporation, Ventana Medical Systems
Get The Full Companion Diagnostics Market Report:
https://www.thebusinessresearchcompany.com/report/companion-diagnostics-global-market-report
Which Region Accounts For The Largest Portion Of The Companion Diagnostics Market?
North America was the largest region in the companion diagnostics market in 2025. The regions covered in the companion diagnostics market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Companion Diagnostics Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/customise?id=7038&type=smp
Browse Through More Reports Similar to the Global Companion Diagnostics Market 2026, By The Business Research Company
Contact Lenses Market Report 2026
https://www.thebusinessresearchcompany.com/report/contact-lenses-global-market-report
Bionic Contact Lenses Market Report 2026
https://www.thebusinessresearchcompany.com/report/bionic-contact-lenses-global-market-report
Smart Contact Lenses Market Report 2026
https://www.thebusinessresearchcompany.com/report/smart-contact-lenses-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model