CT/NG Testing Market: Growth Opportunities Through 2030
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
By 2030, What Market Size Is The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Expected To Reach Based On Its 2026 Value?
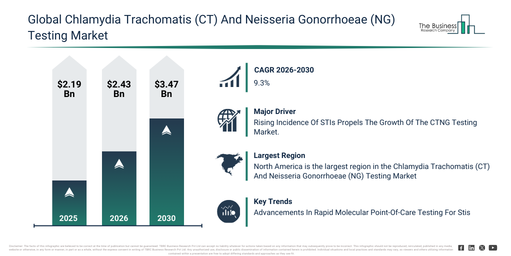
The market size for chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing has experienced rapid expansion in recent times. Projections indicate it will increase from $2.19 billion in 2025 to reach $2.43 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 10.9%. Historically, this growth can be attributed to factors such as enhanced screening for sexually active individuals, the expansion of laboratory testing facilities, the widespread adoption of NAAT and PCR technologies, heightened awareness concerning co-infection risks, and the proliferation of diagnostic laboratories.
The chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing market is projected to experience substantial expansion over the upcoming years. This market is predicted to reach a valuation of $3.47 billion by 2030, driven by a compound annual growth rate (CAGR) of 9.3%. Factors contributing to this anticipated growth include greater investments in decentralized testing, a surge in the embrace of home-based diagnostic methods, the broadening of automated laboratory platforms, an elevated emphasis on prompt STI detection, and an escalating need for comprehensive CT/NG testing solutions. Key developments foreseen during this period involve a rise in the utilization of multiplex STI testing panels, an increasing requirement for swift point-of-care testing, more widespread use of self-testing kits, the development of high-sensitivity molecular assays, and improved integration of laboratory automation.
Access Your Free Sample Report For In-Depth Market Analysis:
Which Strong Drivers Are Impacting The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Growth?
A growing number of sexually transmitted infections (STIs) is anticipated to drive the expansion of the chlamydia trachomatis (CT) and neisseria gonorrhoeae (NG) testing market moving forward. Sexually transmitted infections (STIs) are conditions caused by bacterial, viral, or parasitic infections that can spread through sexual contact. STI rates are increasing due to factors such as inadequate comprehensive sex education, higher sexual activity, social stigma, and various barriers. Chlamydia trachomatis (CT) and neisseria gonorrhoeae (NG) testing is utilized for the detection and management of sexually transmitted infections (STIs), enabling early diagnosis, treatment, and prevention of further transmission. For instance, in October 2025, the Minority HIV/AIDS Fund (MHAF), an US-based government agency, reported that an estimated 39.9 million individuals worldwide were living with HIV in 2023, including 38.6 million adults and 1.4 million children, a figure higher than in previous years. Therefore, the rising incidence of sexually transmitted infections is a key factor propelling the growth of the CTNG testing market.
How Is The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Broken Down By Segment Categories?
The chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing market covered in this report is segmented –
1) By Test Type: Laboratory, Point Of Care Testing
2) By Product: Assays And Kits, Instruments Or Analyzers
3) By Technology: Isothermal Nucleic Acid Amplification Technology, Polymerase Chain Reaction, Immunodiagnostics, Other Technologies
4) By End User: Diagnostic Laboratories, Hospitals And Clinics, Other End Users
Subsegments:
1) By Laboratory: Nucleic Acid Amplification Tests (NAAT), Polymerase Chain Reaction (PCR) Tests, Enzyme-Linked Immunosorbent Assay (ELISA), Immunofluorescence Assay (IFA), Culture Tests, Direct Fluorescent Antibody (DFA) Tests
2) By Point Of Care Testing: Rapid Diagnostic Tests (RDT), Lateral Flow Assays, Immunochromatographic Tests, Self-Testing Kits, Loop-Mediated Isothermal Amplification (LAMP), Microfluidics-Based Tests
Which Trends Are Contributing To Changes In The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market?
Key companies within the chlamydia market are prioritizing the development of innovative solutions, such as the advancement of Point-of-Care Molecular Diagnostics, to achieve quicker, more accurate onsite detection and improve treatment outcomes promptly. This expansion of point-of-care molecular diagnostics involves the wider implementation of rapid, localized molecular tests that enable immediate identification of infections like chlamydia without the need for centralized laboratories. For example, in January 2025, F. Hoffmann-La Roche AG, a Switzerland-based healthcare entity, obtained FDA 510(k) clearance along with a CLIA waiver for its cobas® liat molecular tests, specifically engineered for diagnosing sexually transmitted infections (STIs) directly at the point of care. These tests incorporate multiplex assay panels designed to detect and differentiate various STIs, such as chlamydia, gonorrhea, and Mycoplasma genitalium, from a single patient sample, yielding results in 20 minutes or less.
Which Players Are Present In The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Space?
Major companies operating in the chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing market are F. Hoffmann-La Roche AG, Thermo Fisher Scientific, Abbott Laboratories, Danaher Corporation, Merck KGaA, Siemens Healthineers, Becton Dickinson and Company, Grifols SA, Hologic Inc, PerkinElmer, bioMérieux, Sysmex Corporation, Bio-Rad Laboratories, QIAGEN, Quidel Corporation, DiaSorin, Cepheid, Seegene Inc, Meridian Bioscience Inc, Sekisui Diagnostics, J. Mitra & Co. Pvt. Ltd, Atlas Medical, Hardy Diagnostics, Molbio Diagnostics Pvt. Ltd, NG Biotech
Get The Full Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Report:
Where Is The Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Most Concentrated Geographically?
North America was the largest region in the chlamydia trachomatis (CT) and neisseria gonorrhoeae (NG) testing market in 2025. The regions covered in the chlamydia trachomatis (ct) and neisseria gonorrhoeae (ng) testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Chlamydia Trachomatis (CT) And Neisseria Gonorrhoeae (NG) Testing Market 2026, By The Business Research Company
Chlamydia Infection Diagnostics And Therapeutics Market Report 2026
Chlamydia Infection Diagnostics Market Report 2026
Std Diagnostics Market Report 2026
https://www.thebusinessresearchcompany.com/report/std-diagnostics-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model