Global D Dimer Testing Industry Insights: Key Trends and Growth Estimates
Uncover key drivers, emerging technologies, and competitive movements shaping the d dimer testing market from 2026–2035 with trusted insights from The Business Research Company
How much larger will the D Dimer Testing Market be in 2030 compared with 2026?
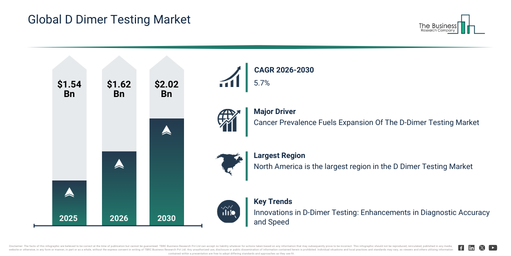
The d dimer testing market has demonstrated significant growth over recent years. It is anticipated to increase from $1.54 billion in 2025 to $1.62 billion in 2026, achieving a compound annual growth rate (CAGR) of 5.2%. This historical expansion can be primarily attributed to factors such as the high incidence of thrombotic disorders, widespread hospital-based laboratory testing, a dependence on elisa methods, the demand for emergency diagnostics, and limited availability of rapid testing options.
The d dimer testing market is anticipated to experience robust growth in the coming years. It is projected to expand to $2.02 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 5.7%. This forecasted growth can be attributed to several factors, including the advancement of point-of-care diagnostics, a rising burden of cardiovascular diseases, technological innovations in immunoassays, the demand for faster turnaround times, and the broader expansion of decentralized testing. Major trends expected during this period include the increasing adoption of point-of-care testing, its enhanced utilization in emergency diagnostics, the proliferation of automated laboratory analyzers, the development of rapid testing kits, and their integration into clinical decision protocols.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12341&type=smp
What Drivers Are Guiding Growth Patterns In The D Dimer Testing Market?
The d-dimer testing market is anticipated to expand due to the rising incidence of cancer. Cancer refers to a group of diseases characterized by the uncontrolled multiplication of abnormal cells in almost any body organ or tissue, which then exceed their normal limits, spreading to other organs or invading adjacent areas. Primarily, d-dimer testing aids in the diagnosis, monitoring, and treatment of cancer patients, while also evaluating the likelihood of blood clot formation. It serves to track thrombotic events across diverse clinical environments. As an illustration, the National Cancer Institute (NCI), a US government entity focused on cancer research and education, projected that in May 2025, approximately 2,041,910 new cancer cases would be diagnosed in the United States during 2025, with 618,120 deaths attributed to the illness. For men, prostate, lung, and colorectal cancers are forecasted to constitute about 48% of all new cancer diagnoses, whereas for women, breast, lung, and colorectal cancers are expected to make up around 51% of all new cases. Furthermore, an estimated 14,690 children and adolescents aged 0 to 19 are predicted to be diagnosed with cancer in 2025, and 1,650 are anticipated to succumb to the condition. Consequently, the rising occurrence of cancer is a key factor driving the expansion of the d-dimer testing market.
How Is The D Dimer Testing Market Divided Into Segments?
The d dimer testing market covered in this report is segmented –
1) By Test: Clinical Laboratory Tests, Point-Of-Care Tests
2) By Product: Analyzers, Reagents And Consumables
3) By Method: Enzyme-Linked Immunosorbent Assay (ELISA), Latex-Enhanced Immuno-Turbidimetric Assays, Fluorescence Immunoassays, Other Testing Methods
4) By Application: Deep Vein Thrombosis (DVT), Pulmonary Embolism (PE), Disseminated Intravascular Coagulation (DIC), Other Applications
5) By End-User: Hospitals, Academic And Research Institutes, Diagnostic Centers, Other End-Users
Subsegments:
1) By Clinical Laboratory Tests: Enzyme-Linked Immunosorbent Assay (Elisa), Chemiluminescent Immunoassay (CLIA), Latex Agglutination Assays
2) By Point-Of-Care Tests: Rapid Test Kits, Portable Testing Devices, Lateral Flow Assays
What Long-Term Trends Are Expected To Shape The Future Of The D Dimer Testing Market?
Major companies operating in the d-dimer testing market are developing innovative products, such as D-dimer test kits, to enhance the accuracy and speed of diagnosing conditions related to blood clots. A D-dimer test kit is a medical diagnostic instrument utilized to measure the concentration of D-dimer, a fibrin degradation product, in the bloodstream, which assists in assessing the existence of blood clots and conditions like deep vein thrombosis, pulmonary embolism, or disseminated intravascular coagulation. For instance, in July 2024, Bionote Inc., a medical equipment manufacturing company located in South Korea, launched Vcheck D-dimer 5Tests/Kit. This new D-dimer 5-Test Kit for Vcheck immunodiagnostic equipment has been specifically designed for veterinary clinics. It helps in diagnosing conditions such as thrombosis and DIC, making it suitable for smaller clinics with fewer emergency cases.
Which Major Industry Participants Are Leading The D Dimer Testing Market Growth?
Major companies operating in the d dimer testing market are F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Abbott Laboratories, Siemens Healthcare GmbH, Becton Dickinson and Company (BD), Laboratory Corporation, Quest Diagnostics Inc., bioMérieux SA, Beckman Coulter Inc., Quidel Corporation, QuidelOrtho Corporation, Sysmex Corporation, Horiba Ltd., Werfen SA, Helena Laboratories Corporation, Abcam plc, LumiraDx Limited, Sekisui Diagnostics LLC, Diagnostica Stago S.A.S., RayBiotech Inc., CTK Biotech Inc., Diazyme Laboratories Inc., UCP Biosciences Inc., AccuBioTech Co Ltd., Response Biomedical Corporation
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/d-dimer-testing-global-market-report
Which Region Is Expected To Experience The Fastest Growth In The D Dimer Testing Market?
North America was the largest region in the D dimer testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the d dimer testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized D Dimer Testing Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=12341&type=smp
Browse Through More Reports Similar to the Global D Dimer Testing Market 2026, By The Business Research Company
Medical Device Testing Market Report 2026
https://www.thebusinessresearchcompany.com/report/medical-device-testing-global-market-report
Dimer Acid Market Report 2026
https://www.thebusinessresearchcompany.com/report/dimer-acid-global-market-report
Dimethyl Ether Market Report 2026
https://www.thebusinessresearchcompany.com/report/dimethyl-ether-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model