Global Decentralized Trial Wearable Market Set for Strong Expansion Reaching $6.18 Billion by 2030 at 20.5% CAGR
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Will Be The Estimated Market Valuation Of The Decentralized Trial Wearable Market By The End Of 2030?
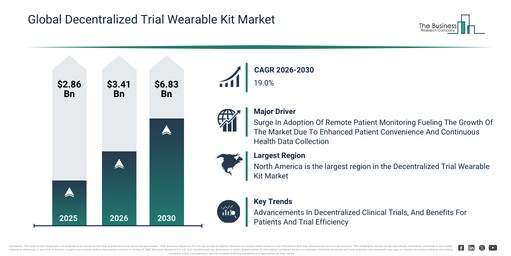
The decentralized trial wearable market size has experienced significant expansion in recent years. It is projected to increase from $2.43 billion in 2025 to $2.93 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 20.8%. The growth observed in the past period is attributable to the increasing adoption of decentralized clinical trial models, the rising use of wearable health devices in research, growing demand for remote patient engagement, advancements in sensor miniaturization, and the expansion of digital clinical trial infrastructure.
The decentralized trial wearable market is projected to experience rapid expansion over the coming years. Its valuation is anticipated to reach $6.18 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 20.5%. This projected growth is driven by several factors, including heightened investments in virtual trial platforms, a surge in demand for generating real-world evidence, the expanding embrace of hybrid clinical trials, a greater focus on remote monitoring that complies with regulations, and the increasing application of AI-driven trial analytics. Key trends anticipated during this period involve the growing acceptance of continuous remote health monitoring wearables, an increase in the integration of multi-parameter biosensing devices, the expanding utilization of real-time data transmission within clinical trials, the broader implementation of patient-centric trial designs, and a heightened commitment to ensuring data accuracy and compliance.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=29012&type=smp
Which Market Drivers Are Supporting The Expansion Of The Decentralized Trial Wearable Market?
The increasing need for real-time patient monitoring is anticipated to fuel the expansion of the decentralized trial wearable market in the foreseeable future. Real-time patient monitoring involves the ongoing gathering and transfer of health data through interconnected medical devices that automatically transmit physiological measurements to healthcare professionals for immediate evaluation and intervention. The need for real-time patient monitoring is escalating due to the growing incidence of chronic conditions such as diabetes, cardiovascular disorders, and respiratory illnesses, which necessitate constant tracking of patients’ health for prompt action and improved disease management. Decentralized trial wearable kits facilitate real-time patient monitoring by continuously collecting and forwarding health data from individuals in their natural settings to central repositories, thereby allowing researchers and healthcare providers to monitor patient health remotely, identify irregularities early, and make prompt, evidence-based clinical choices. For example, in August 2023, Vivalink, a US-based healthcare technology company, reported survey findings indicating that 84% of existing remote patient monitoring (RPM) users intend to increase their usage in 2024, 45% of healthcare providers are deploying RPM for critical monitoring within hospital-at-home programs, and 77% foresee RPM-based care superseding conventional inpatient hospital care within the next five years. Consequently, the rising demand for real-time patient monitoring is a key factor propelling the growth of the decentralized trial wearable market.
Which Segment Classifications Shape The Decentralized Trial Wearable Market?
The decentralized trial wearable market covered in this report is segmented –
1) By Product Type: Smartwatches, Fitness Trackers, Biosensors, Smart Clothing, Other Product Types
2) By Distribution Channel: Online, Offline
3) By Application: Remote Patient Monitoring, Clinical Trials, Disease Management, Other Applications
4) By End-User: Pharmaceutical Companies, Contract Research Organizations, Healthcare Providers, Other End-Users
Subsegments:
1) By Smartwatches: Health Monitoring Smartwatches, Electrocardiogram (ECG) Smartwatches, Blood Oxygen Tracking Smartwatches, Multi-Sensor Smartwatches
2) By Fitness Trackers: Heart Rate Monitoring Fitness Trackers, Activity Tracking Fitness Trackers, Sleep Tracking Fitness Trackers, Multi-Metric Fitness Trackers
3) By Biosensors: Glucose Monitoring Biosensors, Electrocardiogram (ECG) Biosensors, Temperature Sensing Biosensors, Multi-Parameter Biosensors
4) By Smart Clothing: Sensor-Embedded Smart Shirts, Smart Pants With Biometric Sensors, Compression Wear Smart Clothing, Smart Athletic Wear
5) By Other Product Types: Hybrid Wearable Devices, Remote Patient Monitoring Wearables, Medical Grade Wearables, Innovative Material Wearables
What Industry Trends Are Redefining The Decentralized Trial Wearable Market?
Leading companies in the decentralized trial wearable market are concentrating on developing innovative products like ultra-compact health monitoring devices to improve patient comfort and enhance remote data collection. These ultra-compact health monitoring devices are miniature, wearable gadgets designed for continuous tracking of physiological and behavioral data with minimal intrusion, thereby boosting patient compliance, enabling remote supervision, and providing high-quality data for clinical trials. For instance, in June 2025, Empatica Inc., a US-based medical technology firm, introduced EmbraceMini, which is the world’s smallest wrist-worn actigraphy device tailored for clinical research. Measuring just 12mm thick and 14mm wide, a size comparable to a USB flash drive or AA battery, it is engineered for exceptionally comfortable, non-invasive wear, facilitating continuous monitoring of over 200 digital measures, including activity, sleep, gait, and light exposure. The device wirelessly transmits data to Empatica’s FDA-cleared Health Monitoring Platform, supporting remote data acquisition, digital biomarker analysis, and integration with existing clinical trial systems, all while prioritizing participant experience and adherence. It is specifically built for studies on sleep disorders, movement disorders, obesity, depression, and pain, among other conditions, offering high-resolution insights into participants’ physiological and behavioral patterns. Its long battery life allows at least seven days of uninterrupted monitoring, reducing the burden on participants and enhancing compliance. Additionally, the modular design enables it to be worn on the wrist, ankle, leg, or waist, and multiple units can be utilized simultaneously for richer datasets.
Which Companies Are Expanding Their Footprint In The Decentralized Trial Wearable Market?
Major companies operating in the decentralized trial wearable market are Oracle Corporation, IQVIA Holdings Inc., LabCorp Drug Development Inc., ICON Public Limited Company, Garmin Ltd., Clario Inc., Medidata Solutions Inc., BioTelemetry Inc., Signant Health Holding Corp, Withings France SA, Medable Inc., Science 37 Holdings Inc., THREAD Research Inc., Huma Therapeutics Ltd., Empatica S.r.l., Clinical Ink Inc., VivaLNK Inc., Sempulse Ltd., Activinsights Ltd., Mahalo Health Inc.
Read the full decentralized trial wearable market report here:
https://www.thebusinessresearchcompany.com/report/decentralized-trial-wearable-global-market-report
How Is The Decentralized Trial Wearable Market Distributed Across Key Geographic Regions?
North America was the largest region in the decentralized trial wearable market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the decentralized trial wearable market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Decentralized Trial Wearable Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=29012&type=smp
Browse Through More Reports Similar to the Global Decentralized Trial Wearable Market 2026, By The Business Research Company
Virtual Clinical Trials Global Market Report
https://www.thebusinessresearchcompany.com/report/virtual-clinical-trials-global-market-report
Clinical Trial Management System Global Market Report
Clinical Trial Supply And Logistics Global Market Report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model