Diagnostic Contract Manufacturing Market: Drivers and Future Outlook
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
From Its 2026 Market Size, What Value Is The Diagnostic Contract Manufacturing Market Projected To Reach By 2030?
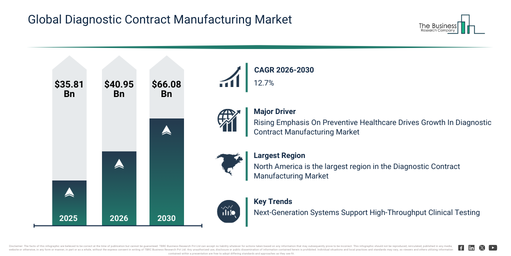
The diagnostic contract manufacturing market has demonstrated rapid expansion in its size over recent years. Forecasts indicate this market will increase from $35.81 billion in 2025 to $40.95 billion in 2026, achieving a compound annual growth rate (CAGR) of 14.4%. This historical growth can be attributed to several factors, including high capital requirements for in-house manufacturing, regulatory complexity, a rise in diagnostic product launches, the demand for manufacturing flexibility, and the globalization of supply chains.
The diagnostic contract manufacturing market is projected to experience substantial expansion over the upcoming years. It is forecast to reach $66.08 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 12.7%. Factors contributing to this growth during the projected period include advancements in diagnostic innovation, the need for quick scale-up capabilities, increased adoption of automation, the expansion of regional manufacturing operations, and a stronger emphasis on robust supply chains. Key trends anticipated during the forecast period involve a rise in the outsourcing of diagnostic manufacturing, a greater call for manufacturing services that adhere to regulatory standards, the broadening of end-to-end CDMO capabilities, an increased emphasis on optimizing costs and ensuring scalability, and the integration of automated and smart manufacturing techniques.
Access Your Free Sample Report For In-Depth Market Analysis:
What Major Factors Are Driving The Diagnostic Contract Manufacturing Market Forward?
The diagnostic contract manufacturing market is anticipated to expand due to a growing emphasis on preventive healthcare. Preventive healthcare encompasses actions designed to avert the onset or progression of ailments, sicknesses, or harm. This heightened focus on prevention stems from escalating healthcare expenditures, evolving healthcare priorities, an increase in chronic conditions, and the aging global population. Diagnostic contract manufacturing plays a role in enhancing the emphasis on preventive healthcare by supplying vital diagnostic instruments and assays, delivering tailored offerings, guaranteeing adherence to quality standards and regulations, facilitating expandability, and allowing for economical approaches to disease prevention and prompt treatment. For example, data from October 2025, released by Eurostat, a Luxembourg-based government agency, indicated that preventive care constituted the subsequent largest category, making up 3.7% of total healthcare spending in the EU during 2023. Within the member states, its proportion was highest in the Netherlands (5.2%) and lowest in Malta (0.9%). Consequently, the growing importance of preventive healthcare is a key factor propelling the expansion of the diagnostic contract manufacturing market.
Which Segment Categories Are Included In The Diagnostic Contract Manufacturing Market Segment Analysis?
The diagnostic contract manufacturing market covered in this report is segmented –
1) By Service: Device Development And Manufacturing Services, Quality Management Services, Packaging And Assembly Services, Other Services
2) By Application: In Vitro Diagnostic Devices, Diagnostic Imaging Devices, Other Applications
3) By End User: Medical Device Companies, Pharmaceutical And Biopharmaceutical Companies, Other End Users
Subsegments:
1) By Device Development And Manufacturing Services: Product Design And Prototyping, Regulatory Affairs And Compliance, Manufacturing And Assembly, Testing And Validation
2) By Quality Management Services: Quality Control And Assurance, Auditing And Inspection Services, Risk Management, Compliance And Certification Support
3) By Packaging And Assembly Services: Primary Packaging, Secondary Packaging, Sterilization Services, Custom Packaging Solutions
4) By Other Services: Supply Chain Management, Inventory Management, Logistics And Distribution, Aftermarket Services
Which Trends Are Impacting The Progress Of The Diagnostic Contract Manufacturing Market?
Leading companies active in the diagnostic contract manufacturing market are emphasizing the creation of advanced solutions, such as one-stop chemiluminescence immunoassay (CLIA) platforms. This strategy aims to boost assay sensitivity, streamline production processes, and support the rising demand for high-throughput and precise diagnostic testing. A one-stop CLIA platform functions as a fully integrated system where a contract manufacturer supplies everything from core raw materials and reagent development to open-system analyzers and large-scale production capacities, a contrast to traditional segmented manufacturing which involves various vendors providing individual components. For instance, in April 2023, Fapon Biotech Inc., an IVD solutions provider from China, introduced its chemiluminescence one-stop solution strategy. This launch included both compact and high-capacity open-system CLIA analyzers, namely Shine i1000 and Shine i2000, alongside a portfolio exceeding 100 CLIA reagents covering biomarkers for tumor, cardiac, infectious disease, thyroid, and inflammation. Designed to help diagnostic companies accelerate product introductions and expand their testing options, this solution provides high-quality raw-material sourcing, flexible reagent-development assistance, reduced per-test expenses, and stronger alignment with local manufacturing requirements.
Who Are The Active Companies Shaping The Diagnostic Contract Manufacturing Market?
Major companies operating in the diagnostic contract manufacturing market are Thermo Fisher Scientific Inc., Abbott Laboratories, Jabil Inc., Flex Ltd., Becton Dickinson and Company (BD), Sekisui Diagnostics LLC, Sanmina Corporation, Celestica Inc., Nipro Medical Corporation, Philips-Medisize Corporation, Plexus Corp, West Pharmaceutical Services Inc., Benchmark Electronics Inc., Bio-Rad Laboratories, QIAGEN N.V., Kimball Electronics Inc., Integer Holdings Corporation, Bio-Techne Corporation, Nolato AB, Nova Biomedical Corporation, Meridian Bioscience Inc., Fujirebio Inc., Biokit S.A., Invetech Pty. Ltd., KMC Systems, Savyon Diagnostics Ltd., Prestige Diagnostics U.K. Ltd., Cenogenic Corporation, Avioq Inc.
Get The Full Diagnostic Contract Manufacturing Market Report:
Which Region Has The Greatest Market Share In The Diagnostic Contract Manufacturing Market?
North America was the largest region in the diagnostic contract manufacturing market in 2025. The regions covered in the diagnostic contract manufacturing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Diagnostic Contract Manufacturing Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Diagnostic Contract Manufacturing Market 2026, By The Business Research Company
Antibody Contract Manufacturing Market Report 2026
Medical Device Contract Manufacturing Market Report 2026
Contract Development Manufacturing Organizations Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model