C. difficile-Associated Diarrhea Treatment Market: Developments Driving Growth
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Are The Market Size Estimates For The Difficile-Associated Diarrhea Treatment Market In 2026 And 2030?
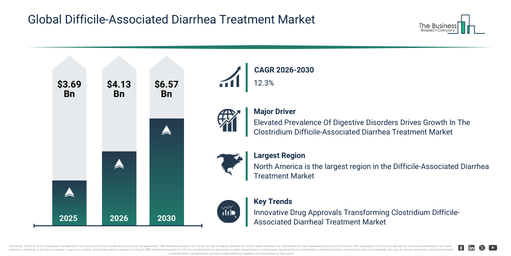
The difficile-associated diarrhea treatment market has experienced substantial expansion in its size over recent years. It is projected to expand from $3.69 billion in 2025 to $4.13 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 11.6%. Factors such as high antibiotic usage rates, the prevalence of hospital-acquired infections, limited treatment options, the vulnerability of the aging population, and prolonged hospital stays contributed to its historical expansion.
The difficile-associated diarrhea treatment market is projected to experience substantial growth in the coming years, anticipated to reach a valuation of $6.57 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 12.3%. Key factors driving this expansion during the forecast period include the introduction of novel antibiotics, various infection control initiatives, strategies aimed at reducing recurrence, wider adoption of improved diagnostics, and the implementation of antimicrobial stewardship programs. Prominent trends expected over the forecast period encompass an increasing preference for narrow-spectrum antibiotics, a heightened emphasis on preventing recurrence, the expansion of treatment protocols within hospitals, growing awareness surrounding antibiotic stewardship, and the advancement of targeted CDI therapies.
Access Your Free Sample Report For In-Depth Market Analysis:
Which Drivers Are Supporting The Difficile-Associated Diarrhea Treatment Market Growth?
The escalating incidence of digestive disorders is projected to boost the expansion of the difficile-associated diarrhea treatment market moving forward. Digestive disorders encompass a range of medical conditions that interfere with the proper functioning of the digestive system, which is responsible for processing food and absorbing nutrients. Clostridium difficile (C. diff) infection is connected to digestive problems, specifically diarrhea and colitis. This bacterium can invade the bowel, generating toxins that lead to inflammation and harm the intestinal lining. With a growing number of individuals diagnosed with digestive disorders, there is an increasing necessity for effective treatments to control associated symptoms like difficile-associated diarrhea. For instance, in June 2023, Crohn’s and Colitis Canada, a non-profit organization based in Canada, reported that an estimated 322,600 Canadians were living with inflammatory bowel diseases (IBD) in 2023, making up approximately 0.82% of the population. Furthermore, projections indicate that around 470,000 Canadians will be affected by IBD by 2035, accounting for about 1.1% of the population, or 1 in every 91 people in the country. Thus, the rising prevalence of digestive disorders is anticipated to stimulate the growth of the difficile-associated diarrhea treatment market.
Which Segment Types Are Examined In The Difficile-Associated Diarrhea Treatment Market Segment Study?
The difficile-associated diarrhea treatment market covered in this report is segmented –
1) By Type: Narrow Spectrum Antibiotics, Broad Spectrum Antibiotics
2) By Distribution Channel: Hospital Pharmacy, Online Pharmacy, Retail Pharmacy
3) By End User: Hospitals, Homecare, Specialty Clinics, Other End-Users
Subsegments:
1) By Narrow Spectrum Antibiotics: Vancomycin, Fidaxomicin, Metronidazole
2) By Broad Spectrum Antibiotics: Ampicillin, Ciprofloxacin, Clindamycin
Which Trends Are Expected To Impact The Difficile-Associated Diarrhea Treatment Market?
Major companies operating in the difficile-associated diarrhea treatment market are focusing on creating and gaining approval for novel drugs to sustain their position in the industry. Drug approvals for difficile-associated diarrhea treatment are on the rise due to the growing need for more effective and safer therapies, exemplified by SER-109, which introduces a new therapeutic choice. For instance, in April 2023, the U.S. Food and Drug Administration, a US-based federal agency, approved SER-109, developed by Seres Therapeutics, a US-based biotech company. This product is the first orally administered fecal microbiota product intended to prevent the recurrence of Clostridioides difficile diarrhea. SER-109 is a live, spore-form probiotic given orally in capsule form, comprising Firmicutes bacteria naturally present in the human gut. Its mechanism involves restoring the bacterial balance in the stomach, which can help prevent C. diff infections from recurring. This medication helps avert the recurrence of Clostridioides difficile (C. diff) infection in individuals aged 18 and above who have completed antibacterial treatment.
Which Players Are Part Of The Competitive Landscape Of The Difficile-Associated Diarrhea Treatment Market?
Major companies operating in the difficile-associated diarrhea treatment market are Pfizer Inc., F. Hoffmann-La Roche AG, Merck & Co. Inc., AbbVie Inc., Sanofi S.A., Teva Pharmaceutical Industries Ltd., Mylan N.V., Sun Pharmaceutical Industries Ltd., Perrigo Company PLC, Dr. Reddy’s Laboratories Ltd., Aurobindo Pharma Limited, Cipla Limited, Hikma Pharmaceuticals plc, Amneal Pharmaceuticals LLC, Zydus Lifesciences Ltd., Lupin Limited, Torrent Pharmaceuticals Ltd., Salix Pharmaceuticals Inc., Seres Therapeutics, Summit Therapeutics, Vedanta Biosciences, Finch Therapeutics Group
Get The Full Difficile-Associated Diarrhea Treatment Market Report:
Which Region Is The Top Contributor To The Difficile-Associated Diarrhea Treatment Market By Share?
North America was the largest region in the difficile-associated diarrhea treatment market in 2025. The regions covered in the difficile-associated diarrhea treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Difficile-Associated Diarrhea Treatment Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Difficile-Associated Diarrhea Treatment Market 2026, By The Business Research Company
Antidiarrhoeals Market Report 2026
https://www.thebusinessresearchcompany.com/report/antidiarrhoeals-global-market-report
Inflammatory Bowel Disease Treatment Market Report 2026
Constipation Treatment Market Report 2026
https://www.thebusinessresearchcompany.com/report/constipation-treatment-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model