Edema clinical trials market developments and opportunities (2026–2030)
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Level Of Market Value Growth Is Predicted For The Edema Clinical Trials Market From 2026 To 2030?
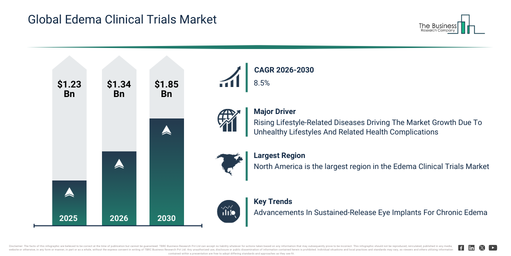
The edema clinical trials market has experienced robust expansion in recent years. Its size is projected to increase from $1.23 billion in 2025 to $1.34 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 8.8%. This historical growth can be attributed to factors such as the rising incidence of cardiovascular and renal disorders, the broadening of clinical research infrastructure, augmented pharmaceutical R&D investments, an increase in investigator-initiated trials, and advancements in clinical trial management systems.
The edema clinical trials market size is projected for robust expansion over the coming years, anticipated to reach $1.85 billion by 2030 at a compound annual growth rate (CAGR) of 8.5%. This growth in the forecast period is attributed to factors such as increasing demand for targeted edema therapies, the rising use of decentralized clinical trial models, the expansion of precision medicine approaches, the growing integration of digital data capture tools, and increased regulatory support for innovative trial designs. Major trends expected within the forecast period include the increasing use of AI-assisted patient recruitment, the rising adoption of adaptive trial designs, a growing focus on biomarker-based endpoints, the expansion of multi-center global trials, and an enhanced emphasis on real-world evidence integration.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/report/edema-clinical-trials-global-market-report
What Key Factors Are Shaping The Edema Clinical Trials Market Landscape?
The rising occurrence of lifestyle-related diseases is anticipated to drive the expansion of the edema clinical trials market in the future. These diseases stem from everyday habits and decisions, such as a sedentary lifestyle, poor dietary choices, and substance consumption. The increase in lifestyle-related diseases is linked to unhealthy eating patterns, where the prevalent intake of processed and high-calorie items contributes to conditions like obesity, diabetes, and cardiovascular issues. Edema clinical trials play a role in addressing lifestyle-related diseases through the creation and evaluation of therapies designed to manage fluid accumulation associated with obesity, diabetes, and cardiovascular conditions. For example, data from March 2024, provided by the Office for Health Improvement & Disparities, a UK government body, indicates that from March 2022 to March 2023, the proportion of individuals with type 1 diabetes receiving all eight suggested care processes saw a 22% increase, and for type 2 diabetes, this figure rose by 21%. Concurrently, the percentage reaching target HbA1c levels climbed to 37.9%, representing the highest recording in the National Diabetes Audit (NDA). Consequently, the growing frequency of lifestyle-related diseases is propelling the expansion of the edema clinical trials market.
How Are Segments Identified Within The Edema Clinical Trials Market Segment Framework?
The edema clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV
2) By Type: Peripheral Edema, Pulmonary Edema, Cerebral Edema, Nephrotic Syndrome Edema
3) By Study Design: Interventional, Observational, Expanded Access
4) By Participant: Pediatrics, Adults, Geriatrics
5) By End User: Pharmaceutical Companies, Research Institutions, Contract Research Organizations (CROs), Academic Research Centers
Subsegments:
1) By Phase I: Single Ascending Dose, Multiple Ascending Dose
2) By Phase II: Proof of Concept, Phase IIb Dose Optimization
3) By Phase III: Pivotal Efficacy Trials, Comparative Trials
4) By Phase IV: Post-Marketing Surveillance, Long-Term Safety Studies
What Market Trends Are Affecting The Edema Clinical Trials Market?
Leading companies active in the edema clinical trials market are concentrating on developing innovative solutions, such as refillable ocular implants, to enhance treatment effectiveness and patient adherence in managing chronic edema-related eye conditions. A refillable ocular implant is defined as a medical device implanted within the eye capable of continuously delivering medication over an extended period, allowing for periodic refilling of the drug without requiring its removal or replacement, thus ensuring sustained treatment for long-term eye conditions like macular edema. For instance, in February 2025, Roche Holding AG, a pharmaceutical company based in Switzerland, obtained approval from the FDA for Susvimo, recognized as the first and only continuous delivery treatment for diabetic macular edema (DME) and diabetic retinopathy (DR), both primary causes of diabetes-related blindness. This offers an alternative to frequent eye injections with significantly fewer annual treatments. This pioneering approval signifies a substantial advancement in ophthalmic care, as Susvimo’s implantable delivery system aids in improving patient compliance and reducing the treatment burden, potentially enhancing long-term outcomes for individuals living with these chronic eye conditions.
Who Are The Companies Competing Within The Edema Clinical Trials Market?
Major companies operating in the edema clinical trials market are Roche Holding AG, Merck & Co. Inc., AbbVie Inc., Bayer AG, Novartis International AG, CSL Limited, Regeneron Pharmaceuticals Inc., Boehringer Ingelheim International GmbH, Genentech Inc., Ionis Pharmaceuticals Inc., ANI Pharmaceuticals Inc., BioCryst Pharmaceuticals Inc., Tactile Medical, REGENXBIO Inc., Koya Medical Inc., KalVista Pharmaceuticals Inc., Kodiak Sciences Inc., Pharvaris B.V., Oxurion NV, Outlook Therapeutics Inc., Adverum Biotechnologies Inc., Oculis Holding AG
Get The Full Edema Clinical Trials Market Report:
https://www.thebusinessresearchcompany.com/report/edema-clinical-trials-global-market-report
Where Is The Edema Clinical Trials Market Primarily Concentrated By Region?
North America was the largest region in the edema clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the edema clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Edema Clinical Trials Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/edema-clinical-trials-global-market-report
Browse Through More Reports Similar to the Global Edema Clinical Trials Market 2026, By The Business Research Company
Virtual Clinical Trials Market Report 2026
https://www.thebusinessresearchcompany.com/report/virtual-clinical-trials-global-market-report
Cardiovascular Clinical Trials Market Report 2026
Healthcare Edi Market Report 2026
https://www.thebusinessresearchcompany.com/report/healthcare-edi-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model