A Significant Growth Driver for the Gene Therapy Clinical Trial Services Market Is the The Increasing Demand For Personalized Medicine Is Fueling The Growth Of The Market Due To Advancements In Genomics And Tailored Therapeutic Approaches
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
How Much Is The Gene Therapy Clinical Trial Services Market Expected To Grow In Terms Of Market Value Between 2026 And 2030?
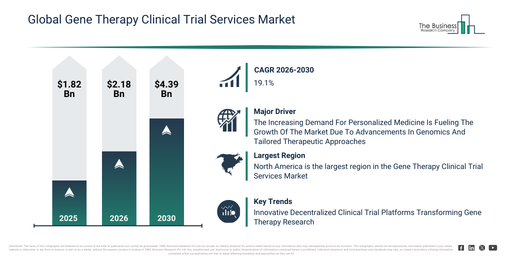
The gene therapy clinical trial services market size has witnessed rapid expansion in recent years. It is forecast to grow from $1.82 billion in 2025 to $2.18 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 19.4%. This historical growth can be attributed to increasing investment in gene therapy R&D, the expansion of early-stage gene therapy pipelines, rising collaboration between biotech firms and CROs, the growing complexity of gene therapy trial protocols, and increased regulatory scrutiny of advanced therapies.
The gene therapy clinical trial services market is projected to experience substantial expansion over the coming years. Its size is anticipated to reach $4.39 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 19.1%. This projected growth during the forecast period can be attributed to several factors, including a rise in late-stage gene therapy trials, a greater need for worldwide trial execution capabilities, the broadening of rare disease gene therapy programs, the increasing adoption of digital trial management platforms, and a heightened emphasis on quicker regulatory approvals. Key trends expected during the same period encompass a surging requirement for specialized management of gene therapy trials, the increasing incorporation of decentralized and hybrid trial models, a heightened concentration on sophisticated cold chain and logistics support, the broadening of patient-centric trial design approaches, and an intensified commitment to regulatory compliance and data integrity services.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=27292&type=smp
What Drivers Are Influencing The Gene Therapy Clinical Trial Services Market?
The growing need for tailored medical treatments is anticipated to drive expansion in the gene therapy clinical trial services market. Personalized medicine is an approach that customizes medical treatment based on individual characteristics, such as genetic, biomarker, and lifestyle factors. The demand for personalized medicine is escalating due to advancements in genomics and biotechnology, which facilitate more precise targeting of therapies. Gene therapy clinical trial services support personalized medicine by analyzing patient-specific genetic profiles and customizing treatments accordingly, utilizing biomarker analysis and companion diagnostics to optimize therapy for each individual while improving safety and effectiveness. For instance, in February 2024, according to the Personalized Medicine Coalition, a US-based hospital and health care organization, in 2023, the FDA approved 16 new personalized treatments for rare disease patients, an increase from six in 2022. Consequently, the increasing demand for personalized medicine is fueling the growth of the gene therapy clinical trial services market.
What Major Segment Divisions Exist Within The Gene Therapy Clinical Trial Services Market?
The gene therapy clinical trial services market covered in this report is segmented –
1) By Type: Clinical Trial Design And Planning, Supply And Logistics Services, Regulatory Services, Data Management And Biostatistics, Site Management And Monitoring, Other Types
2) By Application: Oncology, Hematology, Endocrine or Metabolic Disorders, Musculoskeletal Diseases, Cardiovascular Diseases, Neurology Disorders, Infectious Diseases, Ophthalmology, Immunology, Other Applications
3) By End-User: Pharmaceutical Companies, Biotechnology Companies, Academic Institutions, Government Research Institutes, Contract Research Organizations (CROs)
Subsegments:
1) By Clinical Trial Design And Planning: Protocol Development, Feasibility Studies, Patient Recruitment Strategy Design
2) By Supply And Logistics Services: Cold Chain Management, Investigational Product Packaging And Labeling, Controlled-Temperature Storage
3) By Regulatory Services: Regulatory Consulting And Strategy, Ethics Committee Submissions, Regulatory Compliance Monitoring
4) By Data Management And Biostatistics: Clinical Data Capture And Database Design, Data Validation And Quality Control, Statistical Analysis And Modeling
5) By Site Management And Monitoring: Site Selection And Qualification, Investigator Training And Support, On-Site Monitoring Visits
6) By Other Types: Pharmacovigilance And Safety Reporting, Quality Assurance And Auditing, Patient Engagement And Retention
What Trends Are Advancing Progress In The Gene Therapy Clinical Trial Services Market?
Leading companies in the gene therapy clinical trial services market are concentrating on developing innovative solutions such as the long-term follow-up (LTFU) model for cell and gene therapy (CGT) trials. This effort aims to accelerate the entire trial process while upholding regulatory compliance and safety standards. An LTFU model for cell and gene therapy (CGT) trials is a structured framework designed to monitor patients over an extended duration, frequently up to 15 years after receiving a CGT treatment. For instance, in May 2025, Medable Inc., a US-based SaaS company, unveiled a digital-first Long-Term Follow-Up (LTFU) model specifically engineered for cell and gene therapy (CGT) trials. It supports these trials by reducing burdens on both patients and sites, while simultaneously ensuring scientific rigor throughout the FDA-required 15-year monitoring period. The model enhances patient access, retention, and data continuity through remote and hybrid follow-up methods, thereby improving the monitoring of long-term safety and efficacy.
Which Companies Are Leading Innovation In The Gene Therapy Clinical Trial Services Market?
Major companies operating in the gene therapy clinical trial services market are Thermo Fisher Scientific Inc., Sharp Services LLC, Labcorp, Pharmaceutical Product Development LLC, ICON plc, Syneos Health Inc., WuXi AppTec, Franklin Biolabs, Catalent Inc., Charles River Laboratories International Inc., Parexel International Corporation, Medpace Holdings Inc., Precision Medicine Group LLC, Worldwide Clinical Trials, IQVIA Holdings Inc., TFS HealthScience, Veristat LLC, Novotech Health Holdings Pte Limited, Covance Inc., PPD Clinical Research Services, PRA Health Sciences Inc.
Read the full gene therapy clinical trial services market report here:
Which Regions Are Expected To Experience Rapid Expansion In The Gene Therapy Clinical Trial Services Market?
North America was the largest region in the gene therapy clinical trial services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the gene therapy clinical trial services market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Gene Therapy Clinical Trial Services Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=27292&type=smp
Browse Through More Reports Similar to the Global Gene Therapy Clinical Trial Services Market 2026, By The Business Research Company
Nucleic Acid Based Gene Therapy Global Market Report
Genes Therapy Global Market Report
https://www.thebusinessresearchcompany.com/report/genes-therapy-global-market-report
Cell And Genes Therapy Global Market Report
https://www.thebusinessresearchcompany.com/report/cell-and-genes-therapy-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model