Generic Pharmaceuticals Contract Manufacturing Market Outlook 2026–2030: Key Business Drivers and Trends
Uncover key drivers, emerging technologies, and competitive movements shaping the generic pharmaceuticals contract manufacturing market from 2026–2035 with trusted insights from The Business Research Company
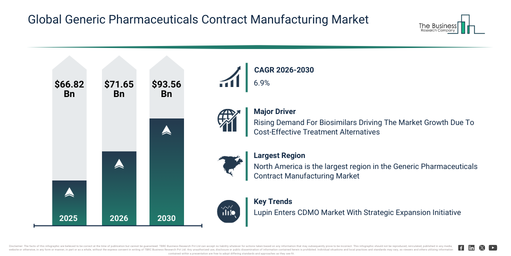
Starting from its 2026 valuation, what market size is the Generic Pharmaceuticals Contract Manufacturing Market expected to reach by 2030?
The generic pharmaceuticals contract manufacturing market size has seen substantial growth recently. This market is projected to expand from $66.82 billion in 2025 to $71.65 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 7.2%. Historically, this expansion has been driven by several factors, including restricted contract manufacturing choices, a past dependence on internal generic drug manufacturing, an increasing need for off-patent active pharmaceutical ingredients (APIs), a rise in generic drug approvals, and financial pressures on pharmaceutical companies.
The market for generic pharmaceuticals contract manufacturing is projected to experience robust expansion over the coming years. This sector is anticipated to reach a valuation of $93.56 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 6.9%. Factors contributing to this expansion during the projection period include an increase in worldwide demand for generic drugs, the growth of contract manufacturing organizations (CMOs), the integration of advanced formulation technologies, a heightened emphasis on biologics and intricate generic products, and enhanced cross-border regulatory partnerships. Key developments expected within this timeframe involve a rise in the outsourcing of generic drug production, an emphasis on economical production and packaging methods, the broadening of services for manufacturing injectables and tablets, greater regulatory assistance and compliance provisions, and an uptick in licensing and technology transfer activities for generic items.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25771&type=smp
What Drivers Are Driving Adoption Within The Generic Pharmaceuticals Contract Manufacturing Market?
The escalating demand for biosimilars is projected to fuel the expansion of the generic pharmaceuticals contract manufacturing market moving forward. Biosimilars are defined as biologic medical products that demonstrate substantial similarity to already approved reference biologics, lacking any clinically relevant distinctions in terms of safety, purity, or potency. The increase in biosimilar demand primarily arises from their capacity to offer economical alternatives to expensive biologic drugs, thereby enhancing the accessibility of advanced treatments for patients and healthcare systems alike. Generic pharmaceuticals contract manufacturing supports biosimilars through the provision of cost-effective, large-scale production capabilities and essential regulatory expertise, which are vital for accelerating market entry and ensuring consistent product quality. For instance, in January 2023, reports from Cardinal Health Inc., a US-based healthcare company, stated that as of January 2022, the U.S. had 33 biosimilar products greenlighted by the FDA, with 21 of them accessible on the market. By 2023, the total number of FDA-approved biosimilars had climbed to 40, with 25 commercially available. Ultimately, the burgeoning demand for biosimilars is serving as a key driver for the growth of the generic pharmaceuticals contract manufacturing market.
How Is The Generic Pharmaceuticals Contract Manufacturing Market Organized Into Various Segments?
The generic pharmaceuticals contract manufacturing market covered in this report is segmented –
1) By Drug Type: Branded Generics, Unbranded Generics
2) By Product Type: Pharmaceutical Active Pharmaceutical Ingredients, Formulations, Capsules, Tablets, Injectables
3) By Route Of Administration: Oral, Parenteral, Topical, Other Routes Of Administration
4) By Application: Oncology, Immunology, Antidiabetic, Neurology, Anticoagulants, Cardiovascular, Respiratory, Pain, Human Immunodeficiency Virus Antivirals
5) By End-User Industry: Pharmaceutical Companies, Contract Research Organizations, Biotechnology Companies, Generic Drug Manufacturers
Subsegments:
1) By Branded Generics: Contract Development of Branded Formulations, Branded Generic Tablet Manufacturing, Branded Injectable Manufacturing, Packaging and Labeling for Branded Generics, Regulatory Support For Branded Generics
2) By Unbranded Generics: Bulk Manufacturing of Off-Patent Active Pharmaceutical Ingredients, Generic Tablet Or Capsule Production, Parenteral Or Injectable Generic Manufacturing, Cost-Efficient Packaging for Unbranded Products, Licensing And Tech Transfer for Generic Products
What Trends Are Expected To Impact The Competitive Landscape Of The Generic Pharmaceuticals Contract Manufacturing Market?
Leading companies in the generic pharmaceutical contract manufacturing market are expanding their service portfolios by forming dedicated subsidiaries to address the rising demand for outsourced development and manufacturing. This strategic initiative of establishing specialized subsidiaries allows organizations to optimize the production of active pharmaceutical ingredients (APIs), ensuring regulatory compliance, scalability, and operational effectiveness throughout the drug development lifecycle. For example, in July 2025, Lupin, an India-based generic pharmaceutical company, unveiled its new subsidiary, Lupin Manufacturing Solutions (LMS), to enter the contract development and manufacturing organization (CDMO) market. This specialized division is focused on supporting the development, manufacturing, and sale of APIs for global pharmaceutical partners. With a leadership team proficient in biotech and pharma, the subsidiary is capable of delivering high-quality contract services, aiding pharmaceutical companies in accelerating their time-to-market while adhering to regulatory standards and cost efficiency.
Who Are The Major Companies Operating In The Generic Pharmaceuticals Contract Manufacturing Market?
Major companies operating in the generic pharmaceuticals contract manufacturing market are WuXi AppTec Co. Ltd., Recipharm AB, Siegfried Holding AG, Curia Global Inc., Piramal Pharma Solutions, Laurus Labs Limited., Cambrex Corp., Alcami Corp. Inc, Neuland Laboratories Limited.
Access The Complete Report For Deeper Market Insights:
Which Region Is Anticipated To See The Fastest Growth In The Generic Pharmaceuticals Contract Manufacturing Market?
North America was the largest region in the generic pharmaceuticals contract manufacturing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the generic pharmaceuticals contract manufacturing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Generic Pharmaceuticals Contract Manufacturing Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=25771&type=smp
Browse Through More Reports Similar to the Global Generic Pharmaceuticals Contract Manufacturing Market 2026, By The Business Research Company
Pharmaceutical Contract Developments And Manufacturing Organization Cmo Market Report 2026
Biopharmaceuticals Contract Manufacturing Market Report 2026
Pharmaceutical Contract Packaging Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model