Global GMP Cell Therapy Consumables Market Trends: Regional Breakdowns and Strategic Insights
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
What Are the Key Milestones in the GMP Cell Therapy Consumables Market’s Growth Trajectory From 2025 To 2034?
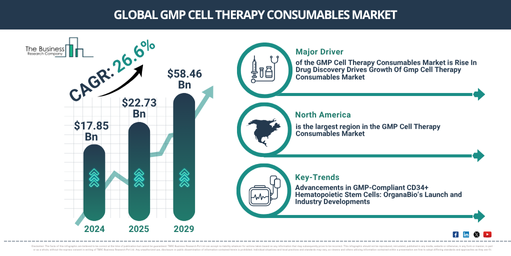
The size of the gmp cell therapy consumables market has seen remarkable growth in recent years. The market is forecasted to expand from a value of $17.85 billion in 2024 to $22.73 billion in 2025, at a compound annual growth rate (CAGR) of 27.3%. Factors such as increased funding in regenerative medicine, progress in cell therapy studies, clinical trials and regulatory approvals, rising instances of chronic ailments and growing understanding of personalized medicine have all contributed to the growth during the historic period.
The market for gmp cell therapy consumables is forecasted to experience remarkable growth in the coming years, reaching an anticipated value of $58.46 billion in 2029 with a compound annual growth rate (CAGR) of 26.6%. The surge expected during the forecast period is primarily attributable to factors such as the broadening application of cell therapy, global upsurge in cell therapy trials, regulatory assistance for cell therapies, emergence of sophisticated cell therapy platforms, and the global expansion of cell therapy manufacturing. Key trends projected for this period encompass investment in manufacturing infrastructure, innovative technology in cell processing, teaming up and forming partnerships, embracing digitalization and data integration, and creating strategic alliances and mergers.
Download a free sample to assess the report’s scope and structure:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12014&type=smp
What Are the Major Market Drivers Behind the Rising Adoption of GMP Cell Therapy Consumables Market?
The increase in drug discovery techniques is anticipated to significantly promote the expansion of the GMP cell therapy consumables market. This is primarily because drug discovery pertains to the process of unveiling new potential medicinal units through the integration of computational, experimental, translational, and clinical models. The evolution and advancement in areas such as molecular biology, genetics, bioinformatics, and high-throughput screening tactics have brought about a major transformation in the drug discovery process. With these technologies, scientists have developed a deeper comprehension of disease mechanisms, pinpointed potential drug targets, and conducted efficient screenings of multiple compound libraries to identify potential drug candidates. This accelerated rate of drug discovery is playing a vital role in the progression of the GMP cell therapy consumables market. As evidence of this, the Food and Drug Administration, a U.S.-based federal organization under the Department of Health and Human Services, reported in January 2023 that the FDA’s Centre for Drug Evaluation and Research (CDER) sanctioned 37 new drugs and therapeutic biological items in the U.S., for diverse indicatives in 2022. Thus, the ascension in drug discovery is serving as a key driver for the GMP cell therapy consumables market’s growth.
Which Key Market Segments Comprise the GMP Cell Therapy Consumables Market and Drive Its Revenue Growth?
The gmp cell therapy consumablesmarket covered in this report is segmented –
1) By Product: Kits, Reagents Or Molecular Biology reagents, Growth Factors Or Cytokines And Interleukins, Other Products
2) By Cell Therapy: NK Cell Therapy, Stem Cell Therapy, T-Cell Therapy, Other cell therapies
3) By Process: Cell Collection And Characterization Or Sorting And Separation, Cell Culture And Expansion Or Preparation, Cryopreservation, Cell Processing And Formulation, Cell Isolation And Activation, Cell Distribution Or Handling, Process Monitoring And Control Or Readministration Or Quality Assurance, Other Processes
4) By End-Use: Clinical, Commercial, Research
Subsegments:
1) By Kits: Cell Culture Kits, Cell Isolation Kits, Transfection Kits
2) By Reagents Or Molecular Biology Reagents: Buffers And Solutions, Enzymes, Staining Reagents
3) By Growth Factors Or Cytokines And Interleukins: Hematopoietic Growth Factors, Antibody Cytokines, Immunomodulatory Cytokines
3) By Other Products: Cell Culture Vessels, Bioreactors, Media And Supplements
Request customized data on this market:
https://www.thebusinessresearchcompany.com/customise?id=12014&type=smp
Which Areas Are Leading Regions in the GMP Cell Therapy Consumables Market Expansion Across the Globe?
North America was the largest region in the GMP cell therapy consumables market in 2024. Asia-pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the GMP cell therapy consumables market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
What Are the Strategic Trends Steering theGMP Cell Therapy Consumables Market Direction?
Leading firms in the GMP cell therapy consumables market are focusing on the creation of GMP-compliant CD34+ hematopoietic stem cells (HSCs) to bolster advanced treatments. These refer to a unique variety of stem cell which bears the CD34 marker and is created under Good Manufacturing Practice (GMP) standards. For example, in April 2024, OrganaBio, an American pharmaceutical firm, introduced HematoPAC-HSC-CB-GMP, a readily available source of GMP-compliant CD34+ hematopoietic stem cells (HSCs), sourced from fresh human umbilical cord blood. Harnessing its vast know-how in cell isolation and GMP manufacturing, OrganaBio is able to yield high quantities of highly viable CD34+ HSCs within 24 hours of harvesting. The purpose of this innovation is to pave the way for the creation of futuristic therapies, especially for blood malignancies and inherited disorders.
View the full report here:
https://www.thebusinessresearchcompany.com/report/gmp-cell-therapy-consumables-global-market-report
How Is the GMP Cell Therapy Consumables Market Conceptually Defined?
GMP cell therapy consumables refer to consumable products and goods produced under good manufacturing practices (GMP) guidelines for cell-based therapies. Consumables and tools for cell culture are made to help researchers grow new cells in culture media for gene therapy, vaccine manufacture, tissue culture, toxicity testing, and drug development.
Purchase the full report and get a swift delivery:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=12014
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model