Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Insights Report: Opportunities and Drivers 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the good manufacturing practice (gmp) services for cell and gene therapy market from 2026–2035 with trusted insights from The Business Research Company
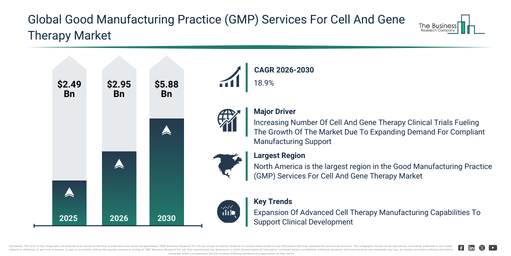
How much is the Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market valued at in 2026, and what valuation is forecast for 2030?
The good manufacturing practice (gmp) services for cell and gene therapy market has seen rapid expansion in recent years. Its size is expected to increase from $2.49 billion in 2025 to $2.95 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 18.6%. This historical growth can be attributed to several factors including the rise in clinical trials for advanced therapies, the broadening of cell therapy research initiatives, more stringent regulatory standardization, the establishment of early-stage gmp facilities, and increased investment in biopharmaceutical manufacturing.
The good manufacturing practice (gmp) services for cell and gene therapy market is anticipated to experience substantial expansion over the upcoming years, projected to reach $5.88 billion by 2030 with a compound annual growth rate (CAGR) of 18.9%. This projected growth throughout the forecast period stems from the increasing commercialization of gene therapies, a heightened demand for scalable manufacturing capacity, the expansion of personalized medicine pipelines, a rise in outsourcing to CDMOs, and the growing adoption of automated quality control systems. Prominent trends identified for this forecast period include the escalating demand for commercial-scale cell and gene therapy manufacturing, the increasing utilization of viral vector production services, a stronger emphasis on GMP-compliant automation, the broadening of fill-finish and cold chain services, and an enhanced focus on regulatory compliance.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=33124&type=smp
What Drivers Are Influencing The Growth Of The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market?
The escalating volume of cell and gene therapy clinical trials is anticipated to drive the expansion of the good manufacturing practice (GMP) services for cell and gene therapy market. These trials represent regulated investigations that evaluate the safety, effectiveness, and quality of advanced therapies, which utilize genetically modified or living cells to treat conditions such as cancer, rare genetic disorders, and autoimmune diseases. The uptick in these trials is fueled by expanded research and development efforts in advanced therapies, increased investment from biotechnology and pharmaceutical companies, and a rising demand for innovative treatments addressing unmet medical needs. Given that many trial sponsors do not possess in-house GMP manufacturing capabilities, they are progressively relying on specialized GMP service providers for compliant manufacturing, analytical testing, quality control, and quality assurance. These particular services are essential for guaranteeing the safe, consistent, and timely delivery of clinical-grade materials across all trial phases. For example, in January 2026, the Bioindustry Association (BIA), a UK-based national trade association, indicated that the UK was conducting 193 advanced therapy clinical trials, with over half in early stages and more than 80% primarily focusing on gene therapies. Thus, the growing number of cell and gene therapy clinical trials is a significant factor boosting the good manufacturing practice (GMP) services for cell and gene therapy market.
How Is The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Organized Into Various Segments?
The good manufacturing practice (gmp) services for cell and gene therapy market covered in this report is segmented –
1) By Service Type: Process Development; Manufacturing; Analytical and Quality Control; Fill-Finish; Other Service Types
2) By Therapy Type: Cell Therapy; Gene Therapy; Combination Therapy
3) By Phase: Preclinical; Clinical; Commercial
4) By End-User: Pharmaceutical and Biotechnology Companies; Academic and Research Institutes; Other End-Users
Subsegments:
1) By Process Development: Vector Design Optimization; Cell Line Development; Upstream Process Optimization; Downstream Purification Development; Process Scale Up Studies
2) By Manufacturing: Clinical Trial Material Production; Commercial Scale Manufacturing; Viral Vector Manufacturing; Plasmid Production Services; Cell Therapy Manufacturing
3) By Analytical and Quality Control: Method Development And Validation; Potency And Efficacy Testing; Safety And Sterility Testing; Stability And Shelf Life Studies; Release Testing Services
4) By Fill-Finish: Aseptic Filling Services; Vial And Syringe Filling; Lyophilization Services; Packaging And Labeling; Cold Chain Handling
5) By Other Service Types: Regulatory Support Services; Technology Transfer Services; Supply Chain Management; Storage And Distribution; Post Manufacturing Support
What Trends Are Reshaping The Dynamics Of The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market?
Leading firms within the good manufacturing practice (GMP) services for cell and gene therapy market are progressively concentrating on enhancing their advanced manufacturing capabilities, such as cell therapy manufacturing, to facilitate regional clinical studies and expedite therapeutic advancement. The production of cell therapies necessitates processes that adhere to GMP standards for the collection, alteration, multiplication, and preparation of therapeutic cells, guaranteeing the product’s safety, quality, and efficacy in clinical settings. A relevant example is AGC Biologics, a US-based contract development and manufacturing organization (CDMO), which in July 2025 introduced services for cell therapy process development and clinical manufacturing at its Yokohama Technical Center in Japan. This facility offers support for GMP-compliant production intended for both preclinical and clinical trials, encompassing CAR-T therapies, induced pluripotent stem cells, and various other stem cell platforms. Such an expansion reinforces AGC Biologics’ worldwide GMP presence across Europe, North America, and Asia, thereby allowing for the quicker and more compliant progression of cell and gene therapies towards commercial availability.
Who Are The Prominent Global Companies Shaping The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Landscape?
Major companies operating in the good manufacturing practice (gmp) services for cell and gene therapy market are Novartis AG, Boehringer Ingelheim BioXcellence GmbH, Batavia Biosciences B.V., FUJIFILM Diosynth Biotechnologies, AGC Biologics Inc., Lonza Group AG, WuXi AppTec Co. Ltd., BioNTech Innovative Manufacturing Services, Charles River Laboratories International Inc., Catalent Inc., Samsung Biologics Co. Ltd., Miltenyi Biotec GmbH, SK pharmteco Co. Ltd., KBI Biopharma Inc., Oxford Biomedica plc, OmniaBio Inc., RoslinCT Ltd., Pluri CDMO, SCTbio Inc., Made Scientific Inc., ENCell Co. Ltd.
Access The Complete Report For Deeper Market Insights:
Which Geographic Areas Are Emerging As Strong Markets For The Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market?
North America was the largest region in the good manufacturing practice (GMP) services for cell and gene therapy market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the good manufacturing practice (gmp) services for cell and gene therapy market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=33124&type=smp
Browse Through More Reports Similar to the Global Good Manufacturing Practice (GMP) Services For Cell And Gene Therapy Market 2026, By The Business Research Company
Cell And Gene Therapy Manufacturing Services Market 2026
Gmp Cell Therapy Consumables Market Report 2026
https://www.thebusinessresearchcompany.com/report/gmp-cell-therapy-consumables-global-market-report
Cell And Gene Therapy Supply Chain Or Logistics Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model