Global Hospital Acquired Infections Testing Kits Market Report 2026–2035 Highlighting Expansion Outlook and Forecast Opportunities
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
How Will The Market Size Of The Hospital Acquired Infections Testing Kits Market Evolve From 2026 To 2030?
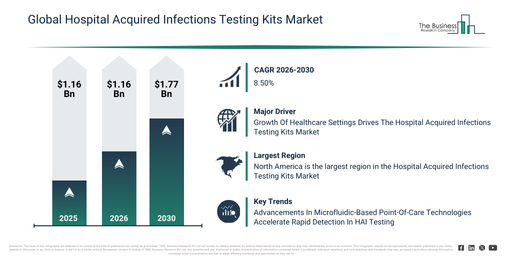
The hospital acquired infections testing kits market size has demonstrated significant growth in recent years. It is anticipated to expand from $4.79 billion in 2025 to $5.19 billion in 2026, reflecting a compound annual growth rate (CAGR) of 8.5%. Historically, this expansion can be ascribed to several factors, including heightened hospital infection surveillance, the escalating prevalence of antibiotic-resistant pathogens, the broadening of clinical microbiology laboratories, the application of infection control protocols, and the accessibility of molecular diagnostic tools.
The market size for hospital acquired infections testing kits is anticipated to demonstrate significant expansion over the next few years, projected to reach $7.2 billion in 2030, exhibiting a compound annual growth rate (CAGR) of 8.5%. This forecasted growth is driven by a heightened focus on early infection detection, increased investments in hospital safety infrastructure, the broader adoption of rapid testing methods, a rising demand for automated diagnostic workflows, and advances in molecular diagnostics technology. Prominent trends within this period include the increased uptake of rapid diagnostic test kits, the growing utilization of molecular testing for HAIs, enhanced integration of automated diagnostic instruments, the expansion of point-of-care infection testing, and a stronger emphasis on multi-pathogen detection.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=2469&type=smp
What Key Drivers Are Influencing The Growth Of The Hospital Acquired Infections Testing Kits Market?
The increasing number of healthcare facilities is projected to drive the expansion of the hospital-acquired infections testing kits market moving forward. Within a healthcare setting, a wide array of facilities and services are available, including acute care hospitals, urgent care centers, rehabilitation centers, nursing homes, and long-term care facilities, all working together to provide comprehensive healthcare solutions. Hospital-acquired infections testing kits are crucial tools for detecting and preventing infections acquired in healthcare environments, thereby enhancing patient safety. For instance, in May 2023, the American Hospital Association, a US-based healthcare industry trade group, reported a significant increase in the total number of hospitals across the U.S. region, rising from 6,093 in 2022 to 60,129 in 2023. Additionally, admissions in community hospitals also grew, climbing from 31,393,318 in 2022 to 31,967,073 in 2023. Thus, the growing presence of healthcare settings is propelling the hospital-acquired infections testing kits market.
Which Segment Areas Are Covered In The Hospital Acquired Infections Testing Kits Market Analysis?
The hospital acquired infections testing kits market covered in this report is segmented –
1) By Product: Consumables And Reagents, Instruments
2) By Test Type: Pneumoia Infection, Urinary Track Infection, Blood Stream Associated Infection, Surgical Site Infection, MRSA infection, Others
3) By Pathogen Type: Viral, Bacterial, Fungal
4) By Application: Drug Resistance Testing, Disease Testing
Subsegments:
1) By Consumables and Reagents: Culture Media (Used for isolating and identifying pathogens), Reagents For Molecular Testing (Includes PCR reagents and kits for detecting specific pathogens), Test Kits (Rapid tests and other kits designed for various infections), Other Consumables (Includes swabs, sample collection devices, and preservation solutions)
2) By Instruments: Automated Analyzers (Equipment used for blood culture analysis and identification of pathogens), Molecular Diagnostic Instruments (Machines used for PCR and other molecular testing methods), Microscopes (Used for the identification of pathogens in samples), Incubators And Growth Chambers (For culturing samples under controlled conditions)
What Key Developments And Trends Are Impacting The Hospital Acquired Infections Testing Kits Market?
Leading companies operating within the hospital-acquired infections (HAI) testing kits market are increasingly concentrating on developing advanced point-of-care antimicrobial susceptibility testing (AST) solutions. These solutions aim to facilitate rapid diagnosis, guide targeted therapy, and effectively combat antimicrobial resistance (AMR). Point-of-care AST technology harnesses proprietary microfluidics to swiftly detect bacterial presence and assess antibiotic effectiveness directly from patient urine samples, thereby minimizing diagnostic delays and enhancing treatment decisions. As an illustration, in June 2023, Sysmex Corporation, a Japan-based diagnostics company, unveiled the world’s first point-of-care testing system in Europe capable of performing antimicrobial susceptibility testing in just 30 minutes. This system, which employs a microfluidic-based AST approach for patients with suspected urinary tract infections, significantly reduces turnaround time and bolsters antimicrobial stewardship in primary care settings by enabling the timely and informed use of antibiotics, consequently supporting global endeavors to curb AMR.
Which Global Companies Are Actively Competing In The Hospital Acquired Infections Testing Kits Market?
Major companies operating in the hospital acquired infections testing kits market are Becton Dickinson and Company, Abbott Laboratories, Cepheid, F. Hoffmann-La Roche Ltd, STERIS plc, QIAGEN, Meridian Bioscience Inc., BioMérieux SA, GE Healthcare, Seegene, SD Biosensor, Fujirebio, Sysmex Corporation, Getein Biotech, Hangzhou Biotest Biotech, AmonMed Biotechnology, Beijing Tigsun Diagnostics, Hunan Lituo Biotechnology, ImmunoScience, Vivacheck Lab
Read the full hospital acquired infections testing kits market report here:
Which Region Is Expected To Register The Fastest Growth In The Hospital Acquired Infections Testing Kits Market?
North America was the largest region in the hospital-acquired infections testing kits market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the hospital acquired infections testing kits market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Hospital Acquired Infections Testing Kits Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=2469&type=smp
Browse Through More Reports Similar to the Global Hospital Acquired Infections Testing Kits Market 2026, By The Business Research Company
Viral Diagnostic Test Kits Market Report 2026
https://www.thebusinessresearchcompany.com/report/viral-diagnostic-test-kits-global-market-report
Infection Control Supplies Market Report 2026
https://www.thebusinessresearchcompany.com/report/infection-control-supplies-global-market-report
Hospital Acquired Infection Control Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model