Point-of-Care Diagnostics Devices And Equipment Market Revenue Growth Expected to Reach $118.72 Billion by 2030 with 13.5% CAGR
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
How Much Is The Point-of-Care Diagnostics Devices And Equipment Market Expected To Grow In Terms Of Market Value Between 2026 And 2030?
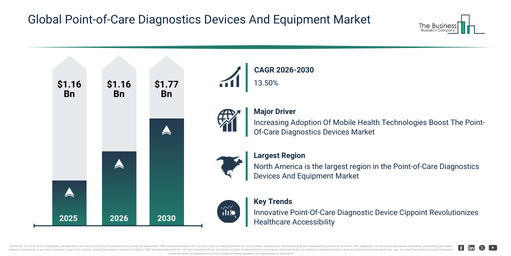
The point-of-care diagnostics devices and equipment market has experienced significant expansion in recent years. This market is forecasted to increase from $64.11 billion in 2025 to $71.62 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 11.7%. Historically, this expansion has been driven by the rising incidence of chronic diseases, the increasing requirement for swift clinical decision-making, the broadening of outpatient healthcare provisions, the growing acceptance of portable diagnostic instruments, and the advancement of preventive healthcare strategies.
The point-of-care diagnostics devices and equipment market is projected to experience substantial expansion over the coming years. This market is set to reach $118.72 billion in 2030, demonstrating a compound annual growth rate (CAGR) of 13.5%. This anticipated growth throughout the forecast period stems from factors such as the increasing need for home healthcare diagnostics, the growing embrace of digital health ecosystems, the broadening of infectious disease screening initiatives, the rising integration of AI-powered diagnostic assistance, and a heightened emphasis on remote patient monitoring. Key developments expected during this period include the wider adoption of rapid diagnostic testing devices, a surging demand for decentralized healthcare diagnostics, the increasing incorporation of connected point-of-care platforms, the proliferation of home-based testing solutions, and a stronger focus on user-friendly device design.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=2387&type=smp
What Key Drivers Are Influencing The Growth Of The Point-of-Care Diagnostics Devices And Equipment Market?
The expanding integration of mobile health innovations is projected to boost the expansion of the point-of-care diagnostics devices market in the future. Mobile health encompasses the utilization of portable devices like smartphones and tablets, alongside other wireless gadgets such as wearables, to provide healthcare services, disseminate information, and oversee health data. Point-of-care diagnostics (POC) devices are vital to mobile health (mHealth) as they facilitate swift and accessible testing and data gathering directly at or close to where the patient is located. Such devices are engineered to deliver instant outcomes for diverse medical examinations and frequently interface with mobile applications or link to smartphones for the purposes of data handling and transfer. For example, PharmiWeb indicated in February2025 that roughly 23% of healthcare interactions are anticipated to occur virtually in 2025. Consequently, the growing acceptance of mobile health technologies is fueling the advancement of the point-of-care diagnostics devices market.
What Major Segment Divisions Exist Within The Point-of-Care Diagnostics Devices And Equipment Market?
The point-of-care diagnostics devices and equipment market covered in this report is segmented –
1) By Type: Infectious disease testing kits, Cardio metabolic monitoring kits, Cholesterol testing kits, Pregnancy and fertility tests kits, Tumor Or cancer markers, Urinalysis testing kits, Hematology testing kits, Drugs of abuse testing kits, Fecal occult testing kits, Other Types
2) By Prescription Mode: Prescription-based Testing Devices, Over-The-Counter (OTC) Testing Devices
3) By End User: Professional diagnostic centers, Hospitals/critical care centers, Outpatient healthcare Setting, Ambulatory care settings, Research laboratories, Home
Subsegments:
1) By Infectious Disease Testing Kits: HIV Testing Kits, Hepatitis Testing Kits, Malaria Testing Kits, Influenza Testing Kits, COVID-19 Testing Kits, Other Infectious Disease Testing Kits
2) By Cardio Metabolic Monitoring Kits: Blood Glucose Testing Kits, Blood Pressure Monitoring Kits, Lipid Testing Kits, Other Cardio Metabolic Monitoring Kits
3) By Cholesterol Testing Kits: Total Cholesterol Testing Kits, HDL Cholesterol Testing Kits, LDL Cholesterol Testing Kits, Triglyceride Testing Kits
4) By Pregnancy and Fertility Test Kits: Pregnancy Test Kits, Ovulation Test Kits, Fertility Monitoring Kits
5) By Tumor Or Cancer Markers: Prostate-Specific Antigen (PSA) Test Kits, Human Epidermal Growth Factor Receptor 2 (HER2) Test Kits, CA-125 Test Kits, Alpha-Fetoprotein (AFP) Test Kits, Other Cancer Marker Test Kits
6) By Urinalysis Testing Kits: Protein Testing Kits, Glucose Testing Kits, pH Testing Kits, Specific Gravity Testing Kits, Nitrite Testing Kits, Other Urinalysis Testing Kits
7) By Hematology Testing Kits: Complete Blood Count (CBC) Kits, Hemoglobin Testing Kits, Hematocrit Testing Kits, Platelet Count Kits, Other Hematology Testing Kits
8) By Drugs of Abuse Testing Kits: Marijuana Testing Kits, Cocaine Testing Kits, Opioid Testing Kits, Amphetamine Testing Kits, Other Drugs of Abuse Testing Kits
9) By Fecal Occult Testing Kits: Stool Blood Test Kits, Occult Blood Screening Kits, Other Fecal Occult Testing Kits
10) By Other Types: Glucose Monitoring Kits, Temperature Monitoring Kits, Oxygen Saturation Monitoring Kits, Other Specialty Point-of-Care Kits
How Are Trends Shaping The Direction Of The Point-of-Care Diagnostics Devices And Equipment Market?
Major companies within the point-of-care diagnostic devices and equipment market are creating innovative products like Cippoint to enhance healthcare access and reduce diagnostic turnaround times. Cippoint serves as an advanced point-of-care testing device applicable to numerous health conditions, offering an extensive array of testing parameters such as cardiac markers, diabetes, infectious diseases, fertility, thyroid function, inflammation, metabolic markers, and coagulation markers. For instance, in January 2023, Cipla Limited, an India-based pharmaceutical company, launched Cippoint, a state-of-the-art point-of-care testing device for multiple health conditions, which also provides a broad spectrum of testing parameters. As an immunofluorescence-based quantitative analyzer, Cippoint enables healthcare professionals to obtain test results in 3 to 15 minutes, thereby facilitating faster and more effective clinical decision-making. Furthermore, it is user-friendly and adaptable for deployment in diverse environments including hospitals, clinics, and mobile vans.
Which Companies Are Leading Innovation In The Point-of-Care Diagnostics Devices And Equipment Market?
Major companies operating in the point-of-care diagnostics devices and equipment market are Abbott Laboratories, F. Hoffmann-La Roche AG, Siemens Healthineers, Danaher, Beckman Coulter, Becton Dickinson and Company, Qiagen N.V., Johnson & Johnson, Alere, BioMerieux, Chembio Diagnostic Systems, Quidel Corporation, EKF Diagnostics, Trinity Biotech Plc, OraSure Technologies, Sysmex Corporation, Radiometer Medical ApS, Response Biomedical Corporation, Bio-Rad Laboratories, Illumina, Thermo Fisher Scientific Inc., LabCorp, Luminex Corporation, LifeScan, Nova, Sekisui Diagnostics, Hemocue AB, Akers Biosciences,
Read the full point-of-care diagnostics devices and equipment market report here:
Which Region Is Expected To Register The Fastest Growth In The Point-of-Care Diagnostics Devices And Equipment Market?
North America was the largest region in the point-of-care diagnostic devices and equipment market in 2025. Asia-Pacific was the second-largest region in the global point-of-care diagnostics devices and equipment market. The regions covered in the point-of-care diagnostics devices and equipment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Point-of-Care Diagnostics Devices And Equipment Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=2387&type=smp
Browse Through More Reports Similar to the Global Point-of-Care Diagnostics Devices And Equipment Market 2026, By The Business Research Company
Point Of Care Diagnostics Market Report 2026
https://www.thebusinessresearchcompany.com/report/point-of-care-diagnostics-global-market-report
Dental Diagnostic Imaging Devices And Equipment Market Report 2026
Hematology Diagnostic Devices And Equipment Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model
[KClientError] [REQ_ERR: OPERATION_TIMEDOUT] [KTrafficClient] Something is wrong.