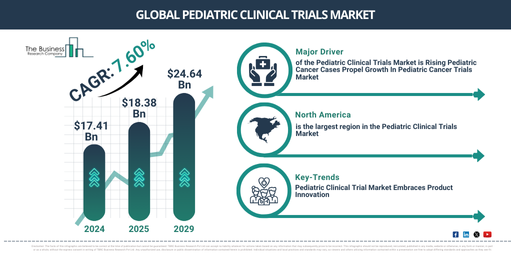
Pediatric Clinical Trials Market Valuation Expected To Reach $24.64 Billion By 2029, Growing At A Rate Of 7.6%
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
What Are the Key Milestones in the Pediatric Clinical Trials Market’s Growth Trajectory From 2025 To 2034?
The market size for pediatric clinical trials has significantly expanded in the past few years. Its growth is projected to continue from $17.41 billion in 2024 to $18.38 billion in 2025, reflecting a compound annual growth rate (CAGR) of 5.6%. Factors driving this growth in the historical period include regulatory incentives, unfulfilled medical requirements, advancements in pediatric medicine, international public health efforts, ethical imperatives, and programs for pediatric vaccination.
The market size of pediatric clinical trials is predicted to experience robust growth in the upcoming years, reaching$24.64 billion in 2029 with a Compound Annual Growth Rate (CAGR) of 7.6%. The growth during this forecast period is attributed to factors like real-world evidence integration, the discovery of pediatric biomarkers, increased access programs, research on pediatric infectious disease, and advancements in pediatric cardiology. The major trends likely to be seen throughout this forecast period encompass patient-focused trial designs, the implementation of adaptive trial designs, digital health technology advancements, worldwide collaboration and consortia, and progress in pediatric oncology.
Download a free sample to assess the report’s scope and structure:
https://www.thebusinessresearchcompany.com/sample.aspx?id=10350&type=smp
Which Factors and External Forces Are Driving Demand in the Pediatric Clinical Trials Market?
Forecasts suggest that the pediatric clinical trials market is likely to expand due to a surge in pediatric cancer incidents. Pediatric cancer, incredibly rare diseases that affect children and teenagers below the age of 14, behaves differently than cancers in adults in terms of growth and dissemination. Pediatric clinical studies remain pivotal in enhancing prognosis and treatment procedures. For example, the American Cancer Society, a voluntary health organization based in the US, projected that roughly 9,910 kids below the age of 15 in the US will receive cancer diagnoses, and around 1,040 of them are anticipated to lose their lives to cancer in 2023. Consequently, this uptick in healthcare spending is fueling the growth of the pediatric clinical trials market.
Which Segments in the Pediatric Clinical Trials Offer the Most Growth?
The pediatric clinical trialsmarket covered in this report is segmented –
1) By Phase: Phase I; Phase II; Phase III; Phase IV
2) By Study Design: Treatment Studies; Observational Studies
3) By Therapeutic Areas: Infectious Diseases; Oncology; Autoimmune Or Inflammatory Diseases; Respiratory Disorders; Mental Health Disorders; Other Therapeutic Areas
Subsegments:
1) By Phase I: First-in-Pediatrics Trials; Dose Escalation Studies; Safety And Tolerability Assessments
2) By Phase II: Efficacy Studies; Dose Optimization Trials; Pharmacokinetic Studies
3) By Phase III: Large-Scale Efficacy Trials; Comparative Studies; Long-Term Safety Studies
4) By Phase IV: Post-Marketing Surveillance; Long-Term Safety Monitoring; Effectiveness Studies In Real-World Settings
Request customized data on this market:
https://www.thebusinessresearchcompany.com/customise?id=10350&type=smp
What Are the Fastest-Growing Geographies in the #Which Factors and External Forces Are Driving Demand in the Pediatric Clinical Trials Market?# Market?
North America was the largest region in the pediatric clinical trials market in 2024. The regions covered in the pediatric clinical trials market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa
Which Cutting-Edge Market Trends Are Expected to Drive thePediatric Clinical Trials Market’s Growth?
The driving trend in the pediatric clinical trial market is product innovation. Firms active in the spectrum of pediatric clinical trials are embracing unique, trailblazing products to solidify their stand in the market. For instance, The Leukemia & Lymphoma Society, a voluntary health organization in the US, introduced the Pediatric Acute Leukemia (PedAL) Master Clinical Trial in June 2022. This trial specifically targets pediatric acute leukemia, using the unique tumor biology of each pediatric cancer patient to align them with the most appropriate and effective treatment.
View the full report here:
https://www.thebusinessresearchcompany.com/report/pediatric-clinical-trials-global-market-report
What Are the Key Elements That Define the Pediatric Clinical Trials Market?
Pediatric clinical trials are trials that investigate the effectiveness, safety, and dosage of pharmaceuticals, medical devices, and other therapies in children. It is essential to develop age-specific, scientifically proven treatments and procedures to identify and improve the best medical therapy possible.
Purchase the full report and get a swift delivery:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=10350
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model