Global Influenza Diagnostic Industry Trends and Market Growth Forecast for 2026–2030
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Market Value Is Expected For The Influenza Diagnostic Market At The End Of The 2026–2030 Forecast Period?
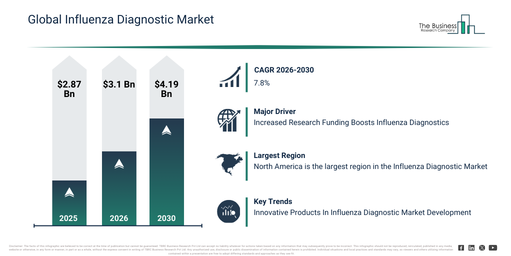
The influenza diagnostic market has demonstrated substantial growth in recent years. This market is expected to expand from $2.87 billion in 2025 to $3.1 billion in 2026, achieving a compound annual growth rate (CAGR) of 8.3%. Historically, this expansion has been propelled by elements like seasonal influenza outbreaks, the extensive adoption of rapid antigen tests, enhancements in hospital laboratory infrastructure, government initiatives for influenza surveillance, and increased recognition of the importance of early flu diagnosis.
The influenza diagnostic market size is projected to experience robust expansion in the upcoming years. By 2030, this market is anticipated to reach $4.19 billion, exhibiting a compound annual growth rate (CAGR) of 7.8%. Factors contributing to this growth during the projected timeframe include an increase in influenza epidemic occurrences, a heightened need for precise molecular diagnostic tools, the broadening of point-of-care testing access, enhanced public health readiness programs, and innovations in quick diagnostic assays. Key trends expected within the forecast period encompass a surge in demand for swift influenza testing, greater integration of molecular diagnostic techniques, a developing preference for point-of-care flu detection, the widening scope of seasonal and pandemic surveillance testing, and ongoing enhancements in the accuracy and reliability of tests.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=3237&type=smp
What Primary Drivers Are Shaping The Development Of The Influenza Diagnostic Market?
The expansion of the influenza diagnostic market is anticipated to be fueled by the growing investment in research concerning influenza diagnostics. This funding refers to financial resources provided by governmental bodies, institutions, and various organizations to back scientific studies aimed at developing, refining, and validating methods and technologies for identifying influenza viruses. The surge in funding for influenza research is primarily motivated by lessons learned from the COVID-19 pandemic and concerns over future respiratory disease outbreaks. Health authorities recognize the crucial importance of rapid and accurate diagnostic capabilities to prevent widespread transmission and enable timely public health interventions. This increased research funding facilitates the development and commercialization of advanced testing technologies, molecular assays, point-of-care devices, and enhanced laboratory procedures, which collectively allow for faster and more accurate identification of influenza strains in clinical settings. For example, in October 2024, the Department of Health and Aged Care, an Australia-based government organization responsible for health research, funding, promotion, and regulation, reported that the Australian Government is dedicating $95 million to combat avian influenza, thereby protecting agriculture, wildlife, and the economy. Thus, the escalating funding for research on influenza diagnostics is a key driver for the growth of the influenza diagnostic market.
What Segment Types Make Up The Influenza Diagnostic Market?
The influenza diagnostic market covered in this report is segmented –
1) By Traditional Diagnostic Tests: Rapid Influenza Diagnostic Tests (RIDT), Viral Culture, DFA, Serological Assays, Other Traditional Diagnostic Tests
2) By Molecular Diagnostic Tests: Reverse Transcription Polymerase Chain Reaction (RT-PCR), Isothermal Nucleic Acid Amplification Tests (INAAT), Loop Mediated Isothermal Based Amplification Assays, Transcription Mediated Isothermal Based Amplification Assays, Other Isothermal Nucleic Acid Amplification Tests, Other Molecular Diagnostic Tests
3) By End User: Hospitals And Clinical Laboratories, Diagnostic Reference Laboratories, Academic/Research Institutes, Other End Users
Subsegments:
1) By Rapid Influenza Diagnostic Tests (RIDT): Immunoassay-based Tests, Lateral Flow Assays, Enzyme Immunoassays (EIA)
2) By Viral Culture: Cell Culture, Egg-based Culture
3) By Serological Assays: Hemagglutination Inhibition (HI) Assay, Enzyme-Linked Immunosorbent Assay (ELISA), Western Blotting Assay
4) By Other Traditional Diagnostic Tests: Polymerase Chain Reaction (PCR), Immunofluorescence Assays
What Major Market Trends Are Driving Changes In The Influenza Diagnostic Market?
Leading companies in the influenza diagnostic market are developing innovative products like the Lucira COVID-19 & Flu Home Test for swift, concurrent qualitative detection. This Lucira COVID-19 and Flu Home Test is a single-use, at-home kit that delivers results from user-collected nasal swab samples in roughly 30 minutes. For example, in February 2023, the US-based pharmaceutical firm Pfizer Inc. received approval for the Lucira COVID-19 & Flu Home Test. The test is capable of detecting influenza A and B, known as the flu, and SARS-CoV-2, the virus responsible for COVID-19.
Who Are The Key Players Driving Competition In The Influenza Diagnostic Market?
Major companies operating in the influenza diagnostic market are F. Hoffmann-La Roche Ltd., Quidel Corporation, Thermo Fisher Scientific Inc., Abbott Laboratories, Hologic Inc., Becton Dickinson and Company, DiaSorin SpA, Luminex Corporation, Meridian Bioscience Inc., GenMark Diagnostics Inc., SA Scientific Ltd., Sekisui Diagnostics LLC, bioMérieux Inc., Danaher Corporation, Siemens Healthineers AG, Coris Bioconcept Sprl, Mast Group, Elitech Technology lnc., Analytik Jena AG, altona Diagnostics GmbH, Tecan Trading AG, Genome Diagnostics Pvt Ltd., Germaine Laboratories Inc., Response Biomedical Corporation, TAUNS Laboratories Inc., TRUPCR Europe Ltd., Quest Diagnostics Inc., Labcorp Employer Services Inc.
Read the full influenza diagnostic market report here:
https://www.thebusinessresearchcompany.com/report/influenza-diagnostic-global-market-report
How Does The Influenza Diagnostic Market Perform Across Major Global Regions?
North America was the largest region in the influenza diagnostics market in 2025. Western Europe was the second largest region in the influenza diagnostics market. The regions covered in the influenza diagnostic market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Influenza Diagnostic Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=3237&type=smp
Browse Through More Reports Similar to the Global Influenza Diagnostic Market 2026, By The Business Research Company
Virtual Clinical Trials Global Market Report
https://www.thebusinessresearchcompany.com/report/virtual-clinical-trials-global-market-report
Clinical Trial Supplies Global Market Report
https://www.thebusinessresearchcompany.com/report/clinical-trial-supplies-global-market-report
Clinical Trial Management Systems Global Market Report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model