Interatrial Shunt Devices Market Anticipated to Record Steady Gains, Advancing to $4.71 Billion by 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the workstation graphics processing unit (gpu) market from 2026–2035 with trusted insights from The Business Research Company
What upper market value could the Interatrial Shunt Devices Market reach by 2030 starting from 2026 levels?
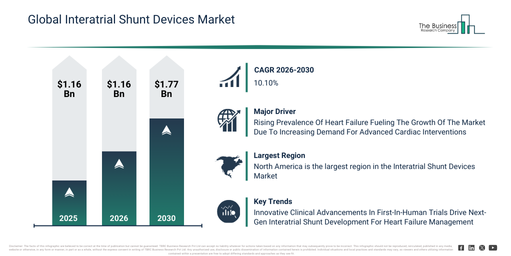
The interatrial shunt devices market has experienced swift expansion in recent years. It is projected to grow from $2.9 billion in 2025 to $3.2 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 10.3%. This historical development can be attributed to the rising prevalence of heart failure, increased understanding of interatrial shunt therapies, advancements in cardiac imaging, a rise in cardiac catheterization procedures, and advantageous reimbursement policies.
The interatrial shunt devices market is anticipated to experience swift expansion over the coming years. By 2030, this market is projected to reach $4.71 billion, demonstrating a compound annual growth rate (CAGR) of 10.1%. Key drivers for this growth during the forecast period include advancements in shunt device technology, broader use of transcatheter delivery techniques, an increasing elderly demographic suffering from cardiovascular ailments, a surge in the preference for minimally invasive interventions, and a heightened emphasis on individualized heart failure treatment. Notable trends emerging in the forecast timeframe encompass minimally invasive cardiac surgeries, tailored heart failure strategies, the shrinking size of devices, an increase in outpatient cardiac services, and sophisticated hemodynamic surveillance.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25798&type=smp
Which Drivers Are Influencing Long-Term Growth In The Interatrial Shunt Devices Market?
The increasing incidence of heart failure is anticipated to fuel the expansion of the interatrial shunt devices market in the future. Heart failure occurs when the heart cannot effectively circulate blood, leading to insufficient delivery of oxygen and nutrients to tissues. The prevalence of heart failure is escalating due to the widespread presence of hypertension, which progressively weakens the heart’s pumping capability. Interatrial shunt devices provide assistance in heart failure by forming a small opening between the left and right atria, enabling the transfer of excess pressure from the overburdened left side of the heart to the right side, thereby mitigating pulmonary congestion and ameliorating symptoms. For instance, in September 2024, the Heart Failure Society of America, a US-based professional organization, reported that as of 2024, approximately 6.7 million Americans aged 20 and above are living with heart failure. This figure is projected to increase to 11.4 million by 2050. Consequently, the growing occurrence of heart failure is propelling the growth of the interatrial shunt devices market.
How Is The Interatrial Shunt Devices Market Divided Into Segments?
The interatrial shunt devices market covered in this report is segmented –
1) By Device Type: Implantable Shunt Devices, Temporary Shunt Devices
2) By Delivery Method: Surgical Delivery, Transcatheter Delivery
3) By Material: Nitinol, Polyester, Polyproplene
4) By Application: Heart Failure With Preserved Ejection Fraction HFpEF, Heart Failure, Left Atrial Pressure Reduction
5) By End-User: Hospitals, Ambulatory Surgical Centers, Cardiac Catheterization Laboratories, Other Users
Subsegments:
1) By Implantable Shunt Devices: Single-Lumen Shunt Devices, Expandable Shunt Devices, Multi-Lumen Shunt Devices
2) By Temporary Shunt Devices: Balloon-Assisted Shunt Devices, Percutaneous Temporary Shunt Devices, Catheter-Based Shunt Devices
What Long-Term Trends Are Expected To Shape The Future Of The Interatrial Shunt Devices Market?
Major companies operating in the interatrial shunt devices market are prioritizing the creation of innovative solutions, exemplified by first-in-human (FIH) clinical trials to evaluate the safety and efficacy of novel shunt technologies for reducing left atrial pressure in heart failure patients. A first-in-human (FIH) clinical trial signifies the initial stage of testing a new medical device or drug in humans, aiming to assess its safety, tolerability, and preliminary efficacy following successful preclinical studies. For example, in January 2025, Adona Medical Inc., a US-based medical technology company, successfully completed patient enrollment for its ATHENS-HF first-in-human (FIH) clinical trial, which investigates a novel interatrial shunt intended for treating heart failure patients. This device is among the pioneers in combining therapeutic shunting with integrated bi-atrial pressure monitoring, aiming to deliver a more adaptive and personalized strategy for heart failure management.
Which Major Firms Are Strengthening Their Position In The Interatrial Shunt Devices Market?
Major companies operating in the interatrial shunt devices market are Abbott Laboratories, Medtronic plc, Terumo Corporation, Edwards Lifesciences Corporation, Cook Medical, Lepu Medical Technology Beijing Co. Ltd., W. L. Gore & Associates Inc., Venus Medtech Inc., Occlutech Holding AG, Wuhan Vickor Medical Technology Co. Ltd., Corvia Medical Inc., Xeltis BV, Alleviant Medical Inc., Adona Medical Inc., Carag AG, InterShunt Technologies Inc., V-Wave Ltd., MicroPort CardioFlow Group Corp., Medinol Ltd., Lifetech Scientific Corp.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/interatrial-shunt-devices-global-market-report
Which Regions Are Poised For Strategic Growth In The Interatrial Shunt Devices Market?
North America was the largest region in the interatrial shunt devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the interatrial shunt devices market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Interatrial Shunt Devices Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=25798&type=smp
Browse Through More Reports Similar to the Global Interatrial Shunt Devices Market 2026, By The Business Research Company
Respiratory Monitoring Devices Global Market Report 2026
Smart Inhalers Global Market Report 2026
https://www.thebusinessresearchcompany.com/report/smart-inhalers-global-market-report
Asthma Drugs Global Market Report 2026
https://www.thebusinessresearchcompany.com/report/asthma-drugs-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
[KClientError] [REQ_ERR: OPERATION_TIMEDOUT] [KTrafficClient] Something is wrong.