Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Anticipated to Record Steady Gains, Advancing to $8.17 Billion by 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the investigational new drug contract development and manufacturing organization (cdmo) market from 2026–2035 with trusted insights from The Business Research Company
By how much is the Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market expected to grow between 2026 and 2030?
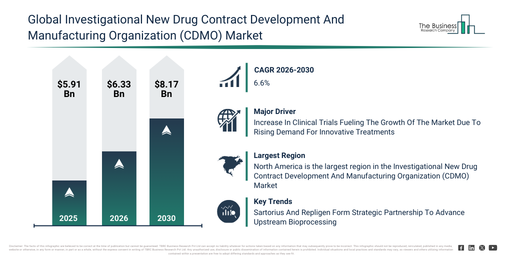
The investigational new drug contract development and manufacturing organization (cdmo) market size has shown substantial growth in recent years. It is forecast to increase from $5.91 billion in 2025 to $6.33 billion in 2026, achieving a compound annual growth rate (CAGR) of 7.1%. The expansion observed in prior periods can be attributed to factors such as the restricted availability of CDMOs for investigational drugs, rising demand for small molecule synthesis, a reliance on conventional manufacturing services, escalating clinical trial volumes, and the early adoption of regulatory support services.
The investigational new drug contract development and manufacturing organization (cdmo) market size is projected to experience robust expansion over the coming years. This market is forecast to reach $8.17 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 6.6%. This anticipated growth during the forecast period stems from factors such as the broadening of gene and cell therapy manufacturing capacities, an increasing need for monoclonal antibodies and recombinant proteins, investments in sophisticated formulation technologies, the implementation of scalable manufacturing platforms, and an increase in worldwide biotechnology research partnerships. Key trends anticipated during this period encompass a heightened demand for drug development services in their early stages, an expansion in services related to the supply of clinical trial materials, a stronger emphasis on adhering to regulatory requirements and quality benchmarks, the proliferation of biologics and advanced therapy development, and the integration of customized manufacturing solutions for both small and large molecules.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25803&type=smp
Which Drivers Are Influencing Market Acceleration In The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
The escalating number of clinical trials is projected to fuel the expansion of the investigational new drug contract development and manufacturing organization (CDMO) market moving forward. Clinical trials themselves are meticulously planned studies with human participants, designed to evaluate the safety and effectiveness of medical treatments, procedures, or interventions under controlled conditions. This uptick in clinical trials is predominantly fueled by a heightened demand for innovative therapies, as the increasing burden of chronic and complex diseases prompts pharmaceutical and biotech companies to develop and evaluate new treatments. Investigational new drug CDMOs play a crucial role in supporting these trials by streamlining the development and manufacturing of trial-ready drug formulations, thereby ensuring a timely and compliant supply of investigational products for each stage of a study. For example, in December 2024, the Association of the British Pharmaceutical Industry, a UK-based pharmaceutical industry trade association, reported that the total number of industry-sponsored clinical trials initiated in the United Kingdom rose from 411 in 2022 to 426 in 2023. Concurrently, Phase III trial initiations saw a 16.5 percent increase over the same period, climbing from 182 to 212. Thus, the growing volume of clinical trials is undeniably a key driver for the investigational new drug contract development and manufacturing organization (CDMO) market’s growth.
What Segments Are Identified Within The Structure Of The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
The investigational new drug contract development and manufacturing organization (cdmo) market covered in this report is segmented –
1) By Product: Small Molecules, Large Molecules
2) By Formulation Type: Solid Formulations, Liquid Formulations, Injectable Formulations
3) By Service Type: Clinical Trial Material Supply, Regulatory Support Services, Manufacturing Services
4) By End-User: Biotechnology Companies, Academic And Research Institutes, Other End-Users
Subsegments:
1) By Small Molecules: Synthetic Small Molecules, Semi-synthetic Small Molecules, Peptide-based Small Molecules, Oligonucleotides
2) By Large Molecules: Monoclonal Antibodies (mAbs), Recombinant Proteins, Vaccines, Cell Therapy Products, Gene Therapy Products, RNA-based Therapeutics
What Trends Are Expected To Impact The Competitive Landscape Of The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
Prominent companies within the investigational new drug contract development and manufacturing organization (CDMO) market are increasingly concentrating on forming strategic partnerships to bolster their development capacities and expedite clinical entry. Strategic partnerships are defined as formal collaborations between two or more entities that cooperate to achieve mutual objectives, leveraging each other’s strengths, resources, or expertise for shared benefits while maintaining their independent status. Illustratively, in August 2023, Sartorius AG, a Germany-based life science company, engaged in a partnership with Repligen Corporation, a US-based life sciences company, to introduce an integrated system that combines BIOSTAT STR and XCell ATF technologies for upstream process intensification. This collaboration is designed to streamline biopharmaceutical production by enhancing both efficiency and scalability. This initiative underscores Sartorius’ continuous innovation in bioprocessing solutions, reaffirming its commitment to advancing the biomanufacturing sector.
Which Major Firms Are Strengthening Their Position In The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
Major companies operating in the investigational new drug contract development and manufacturing organization (cdmo) market are Thermo Fisher Scientific Inc., Fresenius Kabi AG, Lonza Group Ltd., Catalent Inc., Samsung Biologics Co. Ltd., Fareva SA, Recipharm AB, PCI Pharma Services, Vetter Pharma, Delpharm, Kindeva Drug Delivery LP, Ajinomoto Bio-Pharma Services, Cenexi SAS, Grand River Aseptic Manufacturing Inc., INCOG BioPharma Services, Aurigene Pharmaceutical Services, Jubilant HollisterStier LLC, Symbiosis Pharmaceutical Services Ltd., Afton Scientific Corp., Amaran Biotech Inc.
Access The Complete Report For Deeper Market Insights:
Which Region Shows The Strongest Potential For Future Expansion In The Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market?
North America was the largest region in the investigational new drug contract development and manufacturing organization (CDMO) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the investigational new drug contract development and manufacturing organization (cdmo) market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=25803&type=smp
Browse Through More Reports Similar to the Global Investigational New Drug Contract Development And Manufacturing Organization (CDMO) Market 2026, By The Business Research Company
Pharmaceutical Contract Developments And Manufacturing Organization Cmo Market Report 2026
Topical Drugs Contract Development And Manufacturing Organization Cdmo Market Report 2026
Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization Cdmo Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model
[KClientError] [REQ_ERR: OPERATION_TIMEDOUT] [KTrafficClient] Something is wrong.