Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Size and Demand Forecast 2026–2030
Uncover key drivers, emerging technologies, and competitive movements shaping the worker heat stress monitoring wearable market from 2026–2035 with trusted insights from The Business Research Company
How much is the Lateral Flow Immunoassay (LFIA) Based Rapid Test Market valued at in 2026, and what valuation is forecast for 2030?
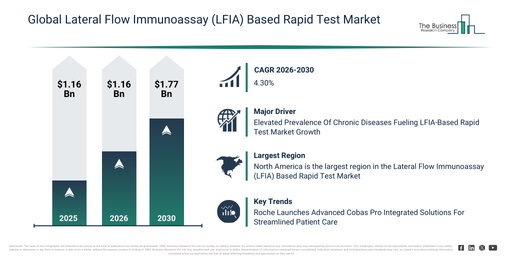
The market for rapid tests utilizing lateral flow immunoassay (LFIA) technology has shown consistent expansion over recent years. This market is projected to expand from $7.17 billion in 2025 to reach $7.31 billion by 2026, exhibiting a compound annual growth rate (CAGR) of 2.0%. Historically, this growth can be attributed to several factors, including the increased scope of rapid infectious disease screening, greater use of pregnancy and fertility tests, the development of point-of-care diagnostics, the presence of affordable rapid tests, and a rise in public health screening programs.
The lateral flow immunoassay (lfia) based rapid test market is projected to experience consistent expansion over the upcoming years. This market is forecast to reach $8.67 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 4.3%. Key factors contributing to this growth during the projection period include escalating investments in decentralized diagnostics, the widening scope of self-testing applications, an increasing need for rapid cardiac and toxicology tests, greater integration with digital health platforms, and an enhanced emphasis on early disease detection. Significant trends anticipated in the forecast timeframe encompass a surge in demand for rapid immunoassay-based testing, the increasing uptake of home-use lfia kits, expanding utilization of multiplex detection assays, broadened applications in infectious disease testing, and an elevated focus on creating user-friendly test designs.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=3293&type=smp
What Drivers Are Influencing The Growth Of The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market?
The escalating prevalence of chronic diseases is projected to stimulate the expansion of the lateral flow immunoassay (LFIA) based rapid test market moving forward. Chronic diseases, also referred to as non-communicable diseases (NCDs), are persistent health conditions that endure over an extended period, frequently lasting a person’s lifetime. Lateral flow immunoassay (LFIA) based rapid tests offer advantages such as early detection, point-of-care testing, rapid results, consistent monitoring, and a reduction in healthcare costs linked to chronic disease care. LFIA-based rapid tests are highly valued in managing chronic diseases due to their capacity to provide prompt and accessible results, enabling patients and healthcare professionals to make timely decisions regarding treatment and dietary adjustments. For instance, in June 2024, according to the National Health Service, a UK-based government department, 3,615,330 individuals registered with a general practitioner (GP) were diagnosed with non-diabetic hyperglycemia or pre-diabetes (a condition with elevated blood sugar levels, not high enough to be classified as diabetes) in 2023, marking an 18% increase from 3,065,825 cases in 2022. Therefore, the rising occurrence of chronic diseases is fueling the growth of the lateral flow immunoassay (LFIA) based rapid test market.
How Is The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Organized Into Various Segments?
The lateral flow immunoassay (lfia) based rapid test market covered in this report is segmented –
1) By Technique: Competitive Assay, Sandwich Assay, Multiplex Detection Assay
2) By Application: Infectious Disease, Pregnancy And Fertility, Toxicology, Cardiac Marker And Cholesterol Testing, Other Applications
3) By End-Users: Hospitals And Clinics, Diagnostic Laboratories, Home Care, Other End Users
Subsegments:
1) By Competitive Assay: Direct Competitive Assays, Indirect Competitive Assays
2) By Sandwich Assay: Single-step Sandwich Assays, Two-step Sandwich Assays
3) By Multiplex Detection Assay: Multi-analyte Assays, Simultaneous Detection Of Various Targets
How Are New Market Trends Shaping The Landscape Of The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market?
Leading companies operating in the lateral flow immunoassay (LFIA)-based rapid test market are concentrating on technologies like Integrated Health Solutions to boost diagnostic effectiveness, streamline operational procedures, and deliver thorough patient care through enhanced data sharing and collaboration across various healthcare platforms. Integrated Health Solutions refer to comprehensive healthcare systems that link diverse services, technologies, and data to improve patient treatment and simplify operations. For instance, in June 2024, F. Hoffmann-La Roche AG, a Switzerland-based pharmaceutical company, introduced the analytical units for its Cobas Pro integrated solutions. The Cobas Pro system is capable of processing up to 1,000 samples per hour with its sample supply unit and provides high-speed analytical units that can perform up to 900 tests per hour for the ISE (ion-selective electrode) and 1,000 tests per hour for the Cobas C 503 unit, which specializes in clinical chemistry.
Who Are The Core Companies Influencing Trends In The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market?
Major companies operating in the lateral flow immunoassay (lfia) based rapid test market are Abbott Laboratories, F. Hoffmann-La Roche, Quidel Corporation, Becton Dickinson and Company, Danaher Corporation, Thermo Fisher Scientific Inc., Bio-Rad Laboratories Inc., PerkinElmer Inc., Hologic Inc., LTEK Co. Ltd., JOYSBIO Biotechnology, Jiangsu Medomics Medical Technologies, Diagnocure, Ubio Biotechnology Systems, Elabscience® Immunoassay Kits, FyoniBio / Glycotope GmbH, Atomo Diagnostics Limited, INDICAL Bioscience, Lionex Gmbh, BIOMED Labordiagnostik GmbH, EKF Diagnostics, Launch Diagnostics, Diagnostic Instruments Ltd, Qiagen N.V., Merck KGaA, 3a Diagnostics, Roche Holding AG, Biomaxima, SensDx, Vitrosens Biotechnology, Bioeasy Biotechnology, Fujirebio, Immunodiagnostic Systems Holdings PLC, Mesa Biotech Inc, CTK Biotech
Access The Complete Report For Deeper Market Insights:
Which Geographic Areas Are Emerging As Strong Markets For The Lateral Flow Immunoassay (LFIA) Based Rapid Test Market?
North America was the largest region in the lateral flow immunoassay (LFIA) based rapid test market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the lateral flow immunoassay (lfia) based rapid test market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Lateral Flow Immunoassay (LFIA) Based Rapid Test Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=3293&type=smp
Browse Through More Reports Similar to the Global Lateral Flow Immunoassay (LFIA) Based Rapid Test Market 2026, By The Business Research Company
Lateral Flow Assays Global Market Report 2026
https://www.thebusinessresearchcompany.com/report/lateral-flow-assays-global-market-report
Immunoassay Global Market Report 2026
https://www.thebusinessresearchcompany.com/report/immunoassay-global-market-report
Immunodiagnostics Global Market Report 2026
https://www.thebusinessresearchcompany.com/report/immunodiagnostics-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company