Top Trends Driving Innovation and Change in the Medical Device Contract Research Organization (CRO) Market: Innovations In Medical Device CRO Market With Rise Of Hybrid And Virtual Trials
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
What Are the Projected Market Size and Growth Rates for the Medical Device Contract Research Organization (CRO) Market From 2025 To 2029?
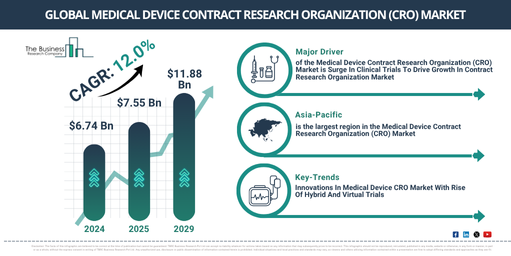
In recent times, the medical device contract research organization (CRO) market has experienced a swift expansion. The market size is projected to rise from $6.74 billion in 2024 to $7.55 billion in 2025, marking a compound annual growth rate (CAGR) of 12.0%. Factors contributing to the growth during the historic period included a growing need for clinical trials, heightened regulatory standards, increased intricacy of medical devices, rising research and development expenditures, and a surge in chronic disease instances.
Over the coming years, there is a predicted swift expansion in the market size of the medical device contract research organization (CRO), potentially reaching a value of $11.88 billion by 2029, reflecting a compound annual growth rate (CAGR) of 12.0%. This projected escalation during the forecasting period can be credited to factors such as the rising acceptance of digital health technologies, heightened concentration on patient safety, proliferation in biologics and biosimilars, increased use of home healthcare devices, and a heightened emphasis on environmental sustainability. Foreseen market trends within this same timeframe include acceptance of AI-driven analytics, widening of personalized medical services, incorporation of real-world evidence into clinical studies, shifting towards patient-centered trial structures, and the addition of cloud-based solutions for data management.
Download a free sample to assess the report’s scope and structure:
https://www.thebusinessresearchcompany.com/sample.aspx?id=19572&type=smp
What External and Internal Drivers Are Contributing to the Growth of theMedical Device Contract Research Organization (CRO) Market?
The upward trend in clinical trials is predicted to instigate the expansion of the medical device contract research organization (CRO) market in the foreseeable future. Clinical trials, the research studies performed on human subjects to assess the safety and efficacy of novel medical treatments, interventions, or diagnostic techniques, have been on the rise. This surge can be attributed to the escalating demand for innovative medical devices and the need to adhere to strict testing rules and regulations. Medical device contract research organizations (CROs) offer special skills and resources for managing, executing, and supervising medical device clinical trials, thereby ensuring compliance and efficient operation. For instance, data from the US-based National Library of Medicine in August 2024 showed that the number of registered clinical trials had grown from 477,227 in 2023 to 506,371 across all US states and 221 countries. Additionally, the library reported registration of 52,731 global medical device studies. Therefore, this rise in clinical trials is set to fuel the development of the medical device contract research organization (CRO) market.
What Segment Types Define the Medical Device Contract Research Organization (CRO) Market Structure?
The medical device contract research organization (cro)market covered in this report is segmented –
1) By Service: Project Management Or Clinical Supply Management, Regulatory Or Medical Affairs, Medical Writing, Clinical Monitoring, Quality Management Or Assurance, Bio Statistics, Investigator Payments, Laboratory, Patient And Site Recruitment, Other Services
2) By Device Type: MedTech Devices, Diagnostic Devices, Handheld Devices, Other Device Types
3) By Phase: Preclinical, Clinical
4) By Application: Cardiology, Diagnostic Imaging, Orthopedic, In Vitro Diagnostics (IVD), Ophthalmic, General And Plastic Surgery, Drug Delivery
Subsegments:
1) By Project Management Or Clinical Supply Management: Study Planning and Coordination, Clinical Supply Chain Management, Vendor Management
2) By Regulatory or Medical Affairs: Regulatory Strategy Development, Medical Affairs Support, Regulatory Submission Preparation
3) By Medical Writing: Clinical Study Protocols, Clinical Study Reports (CSRs), Regulatory Submission Documents, Patient Information Leaflets
4) By Clinical Monitoring: Site Selection and Initiation, On-site Monitoring Visits, Remote Monitoring, Data Verification
5) By Quality Management or Assurance: Quality Assurance Audits, Standard Operating Procedure (SOP) Development, Compliance Management
6) By Biostatistics: Statistical Analysis Plan Development, Data Management And Analysis, Clinical Trial Design
7) By Investigator Payments: Payment Processing Services, Financial Disclosure Management
8) By Laboratory Services: Clinical Laboratory Testing, Bioanalytical Testing, Pathology Services
9) By Patient And Site Recruitment: Site Feasibility Assessment, Patient Recruitment Strategies, Retention And Engagement Strategies
10) By Other Services: Training And Education, Risk Management, Post-Market Surveillance, Data Management Services
Request customized data on this market:
https://www.thebusinessresearchcompany.com/customise?id=19572&type=smp
Which Geographic Areas Hold the Strongest Growth Potential in the Medical Device Contract Research Organization (CRO) Market?
Asia-Pacific was the largest region in the medical device contract research organization (CRO) market in 2024 and is expected to be the fastest-growing region in the forecast period. The regions covered in the medical device contract research organization (CRO) market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
What Long-Term Trends Are Transforming the Competitive Landscape of the Medical Device Contract Research Organization (CRO) Market?
In the medical device contract research organization (CRO) sector, leading businesses are blending conventional services with cutting-edge technologies and enrollment strategies, such as virtual and hybrid trial models, to improve efficiency, accelerate patient participation and simplify regulatory adherence. Catering for various study designs and participant requirements, the functionalities of virtual and hybrid locations are particularly useful with the mounting call for decentralised trials. For example, Lindus Health, a clinical trials firm based in the UK, introduced the All-in-One Medical Device CRO in June 2024. This all-encompassing CRO solution, crafted specially for medical device trials, amalgamates standard CRO services with progressive attributes like their unique Citrus eClinical technology. It combines all crucial trial tools into a single platform, encapsulating clinical trial management systems (CTMS), electronic data capture (EDC) and eConsent. Leveraging its formidable database of over 30 million electronic health records (EHRs) and strategic digital marketing, it improves recruitment for prompt and efficient enrollment. Besides this, Lindus Health delivers robust site services, controlling trials across single-site, multi-site, and virtual/hybrid models through steadfast relationships with chief investigators and research personnel.
View the full report here:
What Is the Definition of the Medical Device Contract Research Organization (CRO) Market?
A medical device contract research organization (CRO) is a specialized service provider that offers outsourced research and development support to medical device manufacturers. These organizations provide a range of services to assist companies in navigating the complex process of bringing a medical device from concept to market, ensuring compliance with regulatory requirements, and achieving successful clinical outcomes. CROs are crucial for reducing the development time, costs, and risks associated with medical device innovations.
Purchase the full report and get a swift delivery:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=19572
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model