Medical Device Safety Testing Market Forecast Highlighting Trends and Growth Potential
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
How Large Will The Medical Device Safety Testing Market Be By 2030 Compared To Its 2026 Market Size?
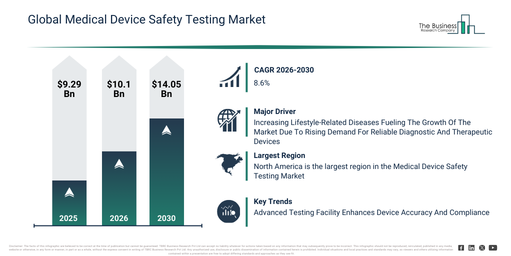
The medical device safety testing market size has experienced robust expansion in recent years. This market is projected to expand from $9.29 billion in 2025 to reach $10.1 billion by 2026, demonstrating a compound annual growth rate (CAGR) of 8.8%. Historically, this growth has been driven by factors such as a rise in medical device recalls, the prompt adoption of international safety standards, an expansion in medical device manufacturing, a heightened emphasis on patient safety, and regulatory oversight by health authorities.
The medical device safety testing market is anticipated to show significant expansion in the coming years. It is projected to achieve a size of $14.05 billion by 2030, growing at a compound annual growth rate (CAGR) of 8.6%. This projected growth can be attributed to several factors including the proliferation of connected and software-driven devices, the tightening of global regulatory frameworks, increasing investment in medical device research and development, a rising demand for expedited market approvals, and the broadening of testing requirements for innovative device technologies. Significant trends for the forecast period include a heightened demand for regulatory compliance testing, an expanding scope for software and cybersecurity testing, the increasing complexity of connected and smart medical devices, a rise in the outsourcing of safety testing services, and the harmonization of global medical device safety standards.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=24296&type=smp
What Leading Drivers Are Supporting The Medical Device Safety Testing Market Expansion?
The rising prevalence of lifestyle-associated illnesses is anticipated to drive the expansion of the medical device safety testing market in the future. These conditions are defined as health issues primarily stemming from an individual’s daily routines and decisions, encompassing ailments such as heart disease, type 2 diabetes, obesity, hypertension, specific cancers, and various mental health concerns. The uptick in lifestyle-related diseases is attributed to insufficient physical activity, which results in increased body weight, compromised cardiovascular health, and diminished insulin sensitivity, thereby elevating the likelihood of chronic illnesses. Medical device safety testing plays a crucial role in managing lifestyle-related diseases by guaranteeing the dependability and operational effectiveness of both diagnostic and therapeutic equipment. This testing method improves patient safety and treatment efficacy by confirming devices adhere to regulatory requirements, consequently fostering greater confidence and streamlined operations within contemporary healthcare systems. As an illustration, in June 2024, data from the National Health Service, a UK-based government body, revealed that 3,615,330 persons registered with a general practitioner (GP) received a diagnosis of non-diabetic hyperglycemia or pre-diabetes in 2023, representing an 18% rise from the 3,065,825 instances recorded in 2022. Consequently, the growing incidence of lifestyle-related diseases is propelling the expansion of the medical device safety testing market.
What Segment Categories Shape The Medical Device Safety Testing Market Segment Landscape?
The medical device safety testing market covered in this report is segmented –
1) By Service Type: Testing, Inspection, Certification
2) By Testing Phase: Preclinical Testing, Clinical Testing
3) By Technology: Active Implantable Devices, In Vitro Diagnostic Devices, Diagnostic Imaging Devices, Ophthalmic Devices
4) By Application: Cardiovascular Devices, Orthopedic Devices, Neurological Devices, Ophthalmic Devices, Dental Devices
5) By End User: Hospitals, Clinics, Research Laboratories, Medical Device Manufacturers, Regulatory Bodies
Subsegments:
1) By Testing: Electrical Safety Testing, Electromagnetic Compatibility (EMC) Testing, Biocompatibility Testing, Environmental Testing, Mechanical Safety Testing, Performance Testing, Software Validation Testing, Chemical Characterization Testing
2) By Inspection: Pre-Market Inspection, In-Process Inspection, Final Product Inspection, Supplier Audit and Evaluation, Manufacturing Site Inspection, Quality System Inspection
3) By Certification: International Organization for Standardization (ISO) 13485 Certification, Conformité Européenne (CE) Marking, Food and Drug Administration (FDA) Premarket Approval (PMA) Support, Medical Device Single Audit Program (MDSAP) Certification, International Electrotechnical Commission (IEC) 60601 Certification, European Union (EU) Medical Device Regulation (MDR) Compliance Certification, Product Safety Certification
What Emerging Trends Are Seen In The Medical Device Safety Testing Market?
Leading companies within the medical device safety testing market are prioritizing expansion through technologically advanced solutions, including sophisticated medical device testing facilities. This focus aims to boost testing accuracy, decrease time-to-market, and ensure adherence to evolving global regulatory standards. An advanced medical device testing facility is a specialized center or laboratory equipped with state-of-the-art technology and expertise to conduct comprehensive evaluation and testing of medical devices. For example, in December 2023, Impelsys, an India-based technology company, inaugurated an advanced medical device testing lab in Mangalore, India. This testing facility employs AI-driven analytics and cloud-based platforms to expedite diagnostic and therapeutic device development through advanced testing, simulations, and regulatory support, thereby demonstrating its commitment to data-driven healthcare innovation.
Who Are The Companies Operating Across The Medical Device Safety Testing Market Value Chain?
Major companies operating in the medical device safety testing market are Thermo Fisher Scientific, Medtronic plc, Baxter International, SGS SA, Eurofins Scientific SE, Bureau Veritas SA, Bausch & Lomb, Intertek Group plc, TÜV SÜD AG, Underwriters Laboratories LLC, Applus+ Group, BSI Group , NSF International , NEMKO AS, Smithers Group Inc., Laboratory Testing Inc., JPen Medical Inc., ArchiMed, F2 Labs Inc., Medistri SA
Get The Full Medical Device Safety Testing Market Report:
https://www.thebusinessresearchcompany.com/report/medical-device-safety-testing-global-market-report
Which Geographic Region Dominates The Medical Device Safety Testing Market?
North America was the largest region in the medical device safety testing market in 2025. Asia-Pacific is expected to be the highest-growing region in the forecast period. The regions covered in the medical device safety testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Medical Device Safety Testing Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/medical-device-safety-testing-global-market-report
Browse Through More Reports Similar to the Global Medical Device Safety Testing Market 2026, By The Business Research Company
Medical Device Technologies Market Report 2026
https://www.thebusinessresearchcompany.com/report/medical-device-technologies-global-market-report
Medical Device Cleaning Market Report
https://www.thebusinessresearchcompany.com/report/medical-device-cleaning-global-market-report
Medical Device Security Market Report
https://www.thebusinessresearchcompany.com/report/medical-device-security-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model