Medical Device Testing Market Performance Outlook 2026–2030: Revenue to Hit $14.22 Billion at 6.4% CAGR
Uncover key drivers, emerging technologies, and competitive movements shaping the medical device testing market from 2026–2035 with trusted insights from The Business Research Company
What growth pattern is forecast for the Medical Device Testing Market size between 2026 and 2035?
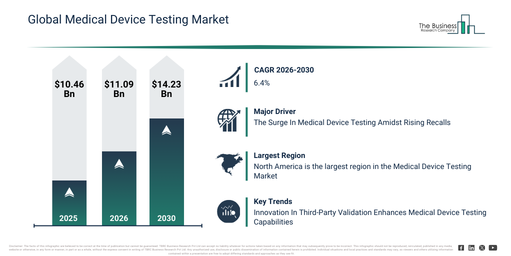
The medical device testing market has experienced robust expansion in recent years. Projections indicate it will expand from $10.46 billion in 2025 to $11.09 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 6.1%. This historical growth is largely due to factors such as an increase in medical device approvals, heightened regulatory oversight, the growing use of implantable devices, the early stages of globalization in medical device manufacturing, and the continuous need for standardized quality assurance.
The medical device testing market is projected to experience substantial expansion over the upcoming years, reaching a valuation of $14.23 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 6.4%. This anticipated growth during the forecast period is primarily driven by factors such as the proliferation of connected medical devices, the increasing intricacy of class III devices, a heightened emphasis on patient safety outcomes, the broadened scope of outsourced testing services, and the dynamic nature of global regulatory frameworks. Key trends characterizing this period are an escalating demand for thorough regulatory compliance testing, a reinforced commitment to device safety and risk mitigation, the expansion of external testing and and certification offerings, a sharpened focus on cybersecurity and software validation assessments, and a greater uptake of sophisticated testing methodologies for high-risk medical devices.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=9981&type=smp
Which Drivers Are Impacting Market Performance In The Medical Device Testing Market?
The medical device testing market is being propelled forward by an increase in medical device recalls. A device recall is a process where the manufacturer or distributor of a device implements corrective actions to remove or rectify an issue within the medical device. Medical device manufacturers use medical device testing to identify potential safety concerns associated with devices, helping to determine if a medical recall is essential to ensure patient safety and device effectiveness, and also improving product quality to prevent recalls. For instance, in March 2024, Sedgwick, a UK-based risk management and brand protection company, reported that 2023 saw 3,306 medical device recalls in Europe (EU + UK), which represented a 20% increase over 2022. Therefore, the rising number of medical device recalls is driving the medical device testing market.
What Segment Types Are Examined In The Medical Device Testing Market?
The medical device testing market covered in this report is segmented –
1) By Testing Type: Physical Testing, Chemical Or Biological Testing, Cybersecurity Testing, Microbiology And Sterility Testing, Other Testing Type
2) By Device Class: Class I, Class II, Class III
3) By Sourcing Type: In-House, Outsourced
4) By Service Type: Testing Services, Inspection Services, Certification Services
5) By End-User: Hospital, Research Center, Homecare Clinics, Other End-User
Subsegments:
1) By Physical Testing: Mechanical Testing, Environmental Testing, Electrical Testing
2) By Chemical Or Biological Testing: Chemical Composition Analysis, Biocompatibility Testing, Toxicological Assessment
3) By Cybersecurity Testing: Vulnerability Assessment, Penetration Testing, Risk Assessment
4) By Microbiology And Sterility Testing: Microbial Contamination Testing, Sterility Assurance Testing, Endotoxin Testing
5) By Other Testing Types: Performance Testing, Usability Testing, Regulatory Compliance Testing
Which Trends Are Guiding The Evolution Of The Medical Device Testing Market?
Leading companies in the medical device testing market are prioritizing innovative approaches to testing and verification, including advanced third-party validation, to guarantee device safety, usability, and regulatory adherence. This advanced third-party validation involves independent testing performed by specialized laboratories to confirm that devices meet performance and regulatory requirements. For example, in October 2023, UL Solutions, a US-based provider of medical testing, inspection, and certification services, introduced expanded medical device testing capabilities at its Rochester Hills laboratory. These new services cover verification and validation, usability testing, cybersecurity assessments, and environmental stress testing, thereby supporting manufacturers in enhancing device safety, reliability, and regulatory compliance.
Who Are The Top-Performing Companies In The Medical Device Testing Market In Recent Years?
Major companies operating in the medical device testing market are Intertek Group plc., SGS SA, Bureau Veritas Group, TUV SUD AG, TUV Rheinland Group, Pace Analytical Services LLC., Charles River Laboratories Inc., UL Solutions, North American Science Associates LLC, Medistri SA, WuXi AppTec Inc., Cordant Health Solutions, GE Healthcare Technologies Inc., Thermo Fisher Scientific Inc., Biomedical Device Labs, NSF International, Analytical Lab Group LLC, Boston Analytical, DDL Inc., Element Materials Technology, Empirical Testing Corp., Eurofins Scientific, Gateway Analytical LLC, iuvo BioScience, Laboratory Corporation of America Holdings, Microtest Laboratories Inc., Nelson Laboratories LLC, National Technical Systems Inc., Pacific Biolabs Inc., Sterigenics International LLC, Toxikon Corporation, WCG Imaging, Westpak Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/medical-device-testing-global-market-report
How Does The Medical Device Testing Market Perform Across Major Global Regions?
North America was the largest region in the medical device testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the medical testing device market report during the forecast period. The regions covered in the medical device testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Medical Device Testing Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=9981&type=smp
Browse Through More Reports Similar to the Global Medical Device Testing Market 2026, By The Business Research Company
Medical Device Engineering Services Market Report 2026
Medical Device Testing Services Market Report 2026
Medical Device Technologies Market Report 2026
https://www.thebusinessresearchcompany.com/report/medical-device-technologies-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model