Medical Devices Vigilance Sector Forecast Shows Robust Momentum, Climbing to $149.02 Billion With 8.8% CAGR
Uncover key drivers, emerging technologies, and competitive movements shaping the medical devices vigilance market from 2026–2035 with trusted insights from The Business Research Company
What long-term size projections exist for the Medical Devices Vigilance Market covering 2026–2035?
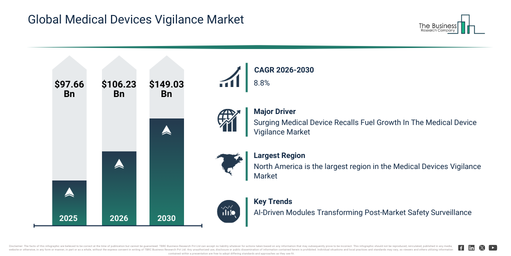
The medical devices vigilance market has seen significant expansion in recent years. Its valuation is forecast to climb from $97.66 billion in 2025 to $106.23 billion in 2026, achieving a compound annual growth rate (CAGR) of 8.8%. This historical growth can be attributed to an increase in reported adverse events linked to devices, the implementation of post-market surveillance regulations, a rise in medical device usage, growing awareness of patient safety, and the broadening of global regulatory frameworks.
The medical devices vigilance market size is anticipated to experience substantial growth over the next few years. It is forecast to expand to $149.03 billion by 2030, achieving a compound annual growth rate (CAGR) of 8.8%. This projected increase during the forecast period is driven by the proliferation of connected and complex medical devices, more rigorous post-market compliance requirements, an uptick in cross-border device approvals, the increasing adoption of proactive risk management, and the necessity for continuous safety monitoring. Prominent trends expected in this period include the reinforcement of post-market surveillance mandates, a greater focus on collecting real-world evidence, intensified regulatory scrutiny on adverse event reporting, the broadening of global vigilance harmonization, and a rising demand for quicker incident response mechanisms.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=14137&type=smp
Which Drivers Are Impacting The Competitive Landscape Of The Medical Devices Vigilance Market?
A rising volume of medical recalls is anticipated to fuel the expansion of the medical device vigilance market moving ahead. Medical recalls pertain to measures undertaken by regulatory bodies, producers, or suppliers to withdraw or rectify medical items that endanger public health or fail to meet regulatory benchmarks. Medical device vigilance involves ongoing observation of medical devices within the market to pinpoint unfavorable incidents, operational failures, or safety worries, which are subsequently communicated to regulatory bodies, possibly prompting additional scrutiny and, if necessary, a product withdrawal. For example, in March 2024, Sedgwick, a UK-based risk management and brand protection company, revealed that in 2023, there were 3,306 medical device recalls in Europe (EU + UK), marking a 20% rise from 2022. Consequently, the growing count of medical recalls is stimulating the expansion of the medical device vigilance market.
What Leading Segments Are Studied In The Medical Devices Vigilance Market?
The medical devices vigilance market covered in this report is segmented –
1) By Delivery Mode: On-Demand, On-Premise
2) By Application: Therapeutics, Diagnostics, Surgical, Research, Other Applications
3) By End-User: Original Equipment Manufacturers (OEMs), Clinical Research Organizations (CROs), Business Process Outsourcing (BPO) Firms
Subsegments:
1) By On-Demand: Cloud-Based Solutions, Subscription Services, Remote Monitoring And Reporting
2) By On-Premise: In-House Software Solutions, Local Server Deployments, Customized System Implementations
Which Trends Are Shaping Activity Within The Medical Devices Vigilance Market?
Leading firms in the medical device vigilance market are prioritizing advancements in post-market safety oversight, particularly through artificial intelligence (AI)-powered modules designed to streamline adverse event reporting, boost operational efficiency, and refine signal identification. These AI-powered modules are essentially smart software elements that leverage artificial intelligence to process, analyze, and rank vigilance information, ensuring regulatory adherence and patient well-being. An example is AB Cube, a medical software firm based in France, which introduced its upgraded customer vigilance platform in July 2024 to assist with the post-market tracking of medical devices. This platform unifies adverse event reporting, trend evaluations, and compliance paperwork within a singular interface. Its operation involves automating the gathering, categorization, and submission of device-related occurrences to regulatory bodies. Distinctive functionalities encompass live analytics dashboards, automated submissions to regulators, and personalized alerts for significant safety events. Its utility extends to medical device manufacturers, notified bodies, and healthcare facilities. The advantages derived are better patient safety, mitigated regulatory exposure, quicker incident resolution, and augmented overall vigilance efficacy.
Which Major Industry Participants Are Leading The Medical Devices Vigilance Market Growth?
Major companies operating in the medical devices vigilance market are Johnson & Johnson, Intel Corporation, Oracle Corporation, Medtronic, Siemens Healthineers, RELX Group plc, MasterControl Inc., Laerdal Medical, Numerix, Smithers, Omnify Software Inc., Freyr, Sparta Systems, MDI Consultants Inc., Greenlight Guru, Jama Software, Sarjen Systems Pvt. Ltd, Arena Solutions Inc., Xybion Corporation, ZEINCRO Group, Extedo Gmbh, AssurX Inc., AB-Cube, Panacea Pharma Projects Limited, Qvigilance, General Electric (GE) Healthcare, Philips Healthcare, Baxter International, Stryker Corporation, Boston Scientific
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/medical-devices-vigilance-global-market-report
Which Geographic Areas Are Emerging As Strong Markets For The Medical Devices Vigilance Market?
North America was the largest region in the medical devices vigilance market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the medical devices vigilance market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Medical Devices Vigilance Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=14137&type=smp
Browse Through More Reports Similar to the Global Medical Devices Vigilance Market 2026, By The Business Research Company
Pharmacovigilance Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharmacovigilance-global-market-report
Patient Monitoring Devices Market 2026
https://www.thebusinessresearchcompany.com/report/patient-monitoring-devices-market
Wearable Medical Devices Market 2026
https://www.thebusinessresearchcompany.com/report/wearable-medical-devices-market
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model