The mRNA Therapeutics CDMO Market Is Evolving Due To Surging Demand For Aesthetic Procedures Fuels Growth In The Botulinum Toxin
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
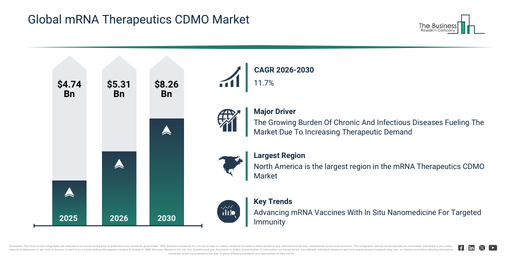
How Much Growth In Market Value Is Expected For The mRNA Therapeutics CDMO Market Between 2026 And 2030?
The mrna therapeutics cdmo market has seen rapid expansion in recent years. It is projected to grow from $4.74 billion in 2025 to $5.31 billion in 2026, at a compound annual growth rate (CAGR) of 12.0%. The market’s historic growth was influenced by the restricted availability of specialized mrna cdmo services, a dependence on in-house pharmaceutical manufacturing, an increase in infectious disease outbreaks, the implementation of early-stage analytical testing, and the broadening of academic and research collaborations.
The mrna therapeutics cdmo market is projected to experience swift expansion over the coming years. By 2030, its valuation is anticipated to reach $8.26 billion, demonstrating a compound annual growth rate (CAGR) of 11.7%. This projected growth is driven by several factors, including increasing investments in mrna therapeutics development, innovations in plasmid DNA and mrna synthesis technologies, the expansion of cancer immunotherapy and protein replacement treatments, a growing need for fill-finish solutions, and the broadening of collaborations between biotechnology firms and CDMOs. Key trends anticipated during this period encompass a surge in the outsourcing of mrna development and production, a heightened emphasis on lipid nanoparticle (LNP) formulation techniques, the enlargement of viral vaccine and cancer immunotherapy portfolios, improved regulatory adherence and quality control protocols, and the wider uptake of analytical and bioanalytical laboratory provisions.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25841&type=smp
What Significant Factors Are Influencing The mRNA Therapeutics CDMO Market Expansion?
The increasing prevalence of chronic and infectious diseases is anticipated to fuel the expansion of the mRNA therapeutics CDMO market in the coming years. These diseases refer to enduring medical conditions like diabetes, cardiovascular disorders, and asthma, alongside illnesses stemming from harmful microorganisms such as viruses, bacteria, and parasites, which can transmit between individuals directly or indirectly. The rise in chronic and infectious diseases is attributable to an aging global population, which is more vulnerable to both prolonged illnesses and infections. The mRNA therapeutics CDMO supports the fight against chronic and infectious diseases by offering comprehensive development and manufacturing services, thus speeding up the delivery of precise, customized mRNA treatments to patients. As an illustration, in October 2024, the World Health Organization, a Switzerland-based international public health organization, reported that around 8.2 million individuals received a new tuberculosis diagnosis in 2023, an increase from approximately 7.5 million in 2022, underscoring the escalating global impact of infectious diseases. Consequently, the rising occurrence of chronic and infectious diseases is a key factor propelling the growth of the mRNA therapeutics CDMO market.
Which Segment Types Are Examined In The mRNA Therapeutics CDMO Market Segment Study?
The mrna therapeutics cdmo market covered in this report is segmented –
1) By Type Of Service: Development Services, Manufacturing Services, Laboratory Services
2) By Indication: Infectious Diseases, Metabolic And Genetic Diseases, Cardiovascular And Cerebrovascular Diseases
3) By Application: Viral Vaccines, Protein Replacement Therapies, Cancer Immunotherapies
4) By End User: Pharmaceutical Companies, Biotechnology Companies, Academic Institutions, Research Organizations
Subsegments:
1) By Development Services: Process Development, Analytical Method Development, Formulation Development, Regulatory Support
2) By Manufacturing Services: Plasmid DNA Manufacturing, mRNA Synthesis, Lipid Nanoparticle (LNP) Formulation, Fill-Finish Services
3) By Laboratory Services: Quality Control Testing, Stability Testing, Bioanalytical Testing, Preclinical Testing
What Emerging Trends Are Seen In The mRNA Therapeutics CDMO Market?
Major companies operating in the mRNA therapeutics CDMO market are prioritizing the development of innovative solutions, such as in situ nanomedicine vaccines, to enhance the effectiveness and precision of mRNA-based vaccines through targeted immune stimulation. An in situ nanomedicine vaccine is a next-generation mRNA-based therapeutic designed to generate nanomedicines within the patient’s body, facilitating targeted delivery and customized immune activation via engineered transgene expression. For instance, in January 2025, Esphera SynBio Inc., a Canada-based biotechnology company, launched a new project supported by the CQDM Quantum Leap program. This initiative aims to improve first-generation lipid nanoparticle or messenger RNA (LNP/mRNA) vaccines by incorporating proprietary transgene designs that induce in vivo production of immunostimulatory nanomedicines. This approach enables precise antigen delivery and tailored immune responses, positioning the technology as a potential breakthrough in vaccine innovation.
Which Players Are Part Of The Competitive Landscape Of The mRNA Therapeutics CDMO Market?
Major companies operating in the mrna therapeutics cdmo market are Thermo Fisher Scientific Inc., Danaher Corporation, Merck KGaA, FUJIFILM Holdings Corporation, Evonik Industries AG, AGC Biologics GmbH, Eurofins Scientific SA, Samsung Biologics Co. Ltd., Wuxi Biologics, Recipharm AB, EMERGENT BioSolutions Inc., Genscript Biotech, Ginkgo Bioworks Holdings Inc., Corden Pharma Corp., TriLink BioTechnologies LLC, Biocina Pty Ltd., Aldevron LLC, Synbio Technologies LLC, etherna manufacturing, Bio-Synthesis Inc., Biomay AG.
Get The Full mRNA Therapeutics CDMO Market Report:
https://www.thebusinessresearchcompany.com/report/mrna-therapeutics-cdmo-global-market-report
Which Region Holds The Highest Market Share In The mRNA Therapeutics CDMO Market?
North America was the largest region in the mRNA therapeutics CDMO market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the mrna therapeutics cdmo market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized mRNA Therapeutics CDMO Market Report For Competitive Insights:
https://www.thebusinessresearchcompany.com/report/mrna-therapeutics-cdmo-global-market-report
Browse Through More Reports Similar to the Global mRNA Therapeutics CDMO Market 2026, By The Business Research Company
Mrna Therapeutics Market Report 2026
https://www.thebusinessresearchcompany.com/report/mrna-therapeutics-global-market-report
Mrna Cancer Vaccines And Therapeutics Market Report 2026
Peptide And Oligonucleotide Cdmo Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at: marketing@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model