What Will The Mycoplasma Testing Market Look Like In 2023?
The Business Research Company’s global market reports are now updated with the latest market sizing information for the year 2023 and forecasted to 2032
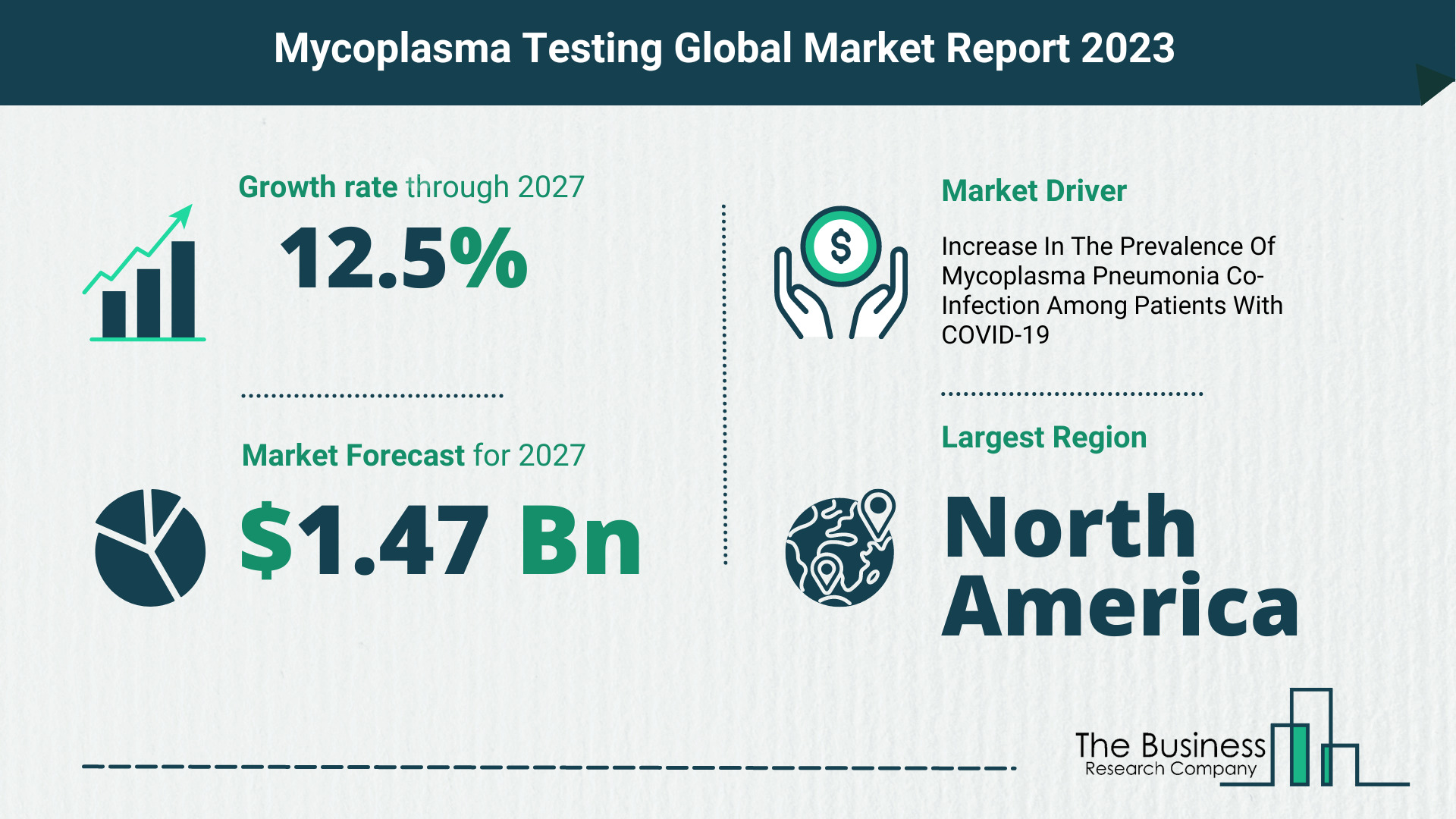
The Business Research Company’s mycoplasma testing market report forecasts the mycoplasma testing market size to grow to $1.47 Billion by 2027, with a CAGR (compound annual growth rate) of more than 12%.
Learn More On The Mycoplasma Testing Market Report 2023 – https://www.thebusinessresearchcompany.com/report/mycoplasma-testing-global-market-report
Mycoplasma Testing Market Size Forecast
The global mycoplasma testing market is expected to grow from $0.81 billion in 2022 to $0.92 billion in 2023 at a compound annual growth rate (CAGR) of 13.1%. The Russia-Ukraine war disrupted the chances of global economic recovery from the COVID-19 pandemic, at least in the short term. The war between these two countries has led to economic sanctions on multiple countries, a surge in commodity prices, and supply chain disruptions, causing inflation across goods and services and affecting many markets across the globe. The mycoplasma testing market size is expected to grow to $1.47 billion in 2027 at a CAGR of 12.5%.
North America held the largest mycoplasma testing market share, and Asia-Pacific was the fastest-growing region in 2022.
Key Mycoplasma Testing Market Driver – Increase In The Prevalence Of Mycoplasma Pneumonia Co-Infection Among Patients With COVID-19
For instance, according to a 2020 study published in the Archives of Medical Laboratory Sciences, the co-infection due to M. pneumonia varied between 1 and 40.6% depending on the population and diagnostic tests used and the combined prevalence of M. pneumoniae co-infection among patients with COVID-19 was reported by 17%.
Request for A Sample Of The Global Mycoplasma Testing Market Report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=4103&type=smp
Key Mycoplasma Testing Market Trend – Undertaking Various Strategic Investments
Major players operating in the market are undertaking various strategic investments such as the launch of innovative products, research and development advancements, and technological innovations, which are gaining significant popularity in the mycoplasma testing market. Companies are introducing new innovative products to meet the requirements of end-users and expand the consumer base to survive in the competitive business environment. For instance, in July 2020, bioMérieux, a France-based biotechnology company introduced Bio Fire Mycoplasma, an advanced test used for bio-therapeutics including hormones, antibodies, gene, and cell therapies for mycoplasma detection in pharmaceutical products.
Mycoplasma Testing Market Segment
1) By Product: Instruments, Assay, Kits, and Reagents
2) By Technology: PCR, ELISA, Enzymatic Methods, DNA Staining, Other Technologies
3) By Application: Cell line Testing, Virus Testing, End- of- Production Cell Testing
4) By End User: Pharmaceutical & Biotechnology Companies, Cell Banks & Laboratories, Contract Research Organizations, Academic Research Institutes
Mycoplasma Testing Market Major Players and Strategies
Major players in the mycoplasma testing market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza, PromoCell GmbH, American Type Culture Collection, Charles River Laboratories International Inc., Bionique Testing Laboratories Inc., Biological Industries Israel Beit Haemek Ltd., InvivoGen, Agilent Technologies, Abbott, Roche Diagnostics, Boston Scientific Corporation, Clongen Laboratories LLC, Creative Bioarray, Nelson Laboratories Fairfield Inc., Norgen Biotek Corporation, Biotools B & M Labs S.A, GenBio, GeneCopoeia Inc., Minerva Biolabs GmbH, Mycoplasma Experience, Savyon Diagnostics, ScienCell Research Laboratories Inc., AngioDynamics, AtriCure Inc., CONMED Corporation, Olympus Corporation, Smith & Nephew, and Advanced Cardiac Therapeutics.
In January 2020, Charles River Laboratories International, Inc., a USA-based company specializing in a variety of preclinical and clinical laboratory services for the biotechnology, pharmaceuticals, and medical devices industry acquired HemaCare Corporation for approximately $380 million in cash. With the acquisition, Charles River Laboratories International, Inc. expands the scientific capabilities of the company in the high-growth cell therapy sector and HemaCare cellular products improve Charles River Laboratories ability to offer a comprehensive cell therapy solution, including mycoplasma detection. HemaCare Corporation is a USA-based company engaged in the production of human-derived biological products and services for cellular therapy development, drug discovery, and biomedical research.
The Mycoplasma Testing Global Market Report 2023 covers regional data on mycoplasma testing market size, mycoplasma testing market trends and drivers, opportunities, strategies, and mycoplasma testing market competitor analysis. The countries covered in the mycoplasma testing market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Russia, South Korea, the UK, and the USA, and the major seven regions are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, the Middle East, and Africa.
The mycoplasma testing detects mycoplasma pneumoniae, which causes mycoplasma pneumonia. Mycoplasma pneumonia is a contagious respiratory infection that spreads easily through contact with respiratory fluids. It can cause epidemics. Mycoplasma testing includes a group of tests that measures antibodies in the blood produced in response to a mycoplasma infection and detects the microbe directly through detecting or culturing its genetic material (DNA) in a body sample.
View More Reports Related To The Mycoplasma Testing Market –
Cold Plasma Global Market Report 2023
Healthcare Analytical Testing Services Global Market Report 2023
Automotive Testing, Inspection And Certification Global Market Report 2023
Contact us at:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 3156230293
Asia +44 2071930708
Europe +44 2071930708
Email us at [email protected]
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model
Found this article helpful? Share it on: