Global Neonatal Point Of Care Testing Market Analysis 2025–2029: Opportunities for Strategic Growth
Unlock Global Insights – Save 20% on Market Reports with Code ONLINE20
How Much Larger Is The Neonatal Point Of Care Testing Market Expected To Become By 2029 Compared With Its 2025 Level?
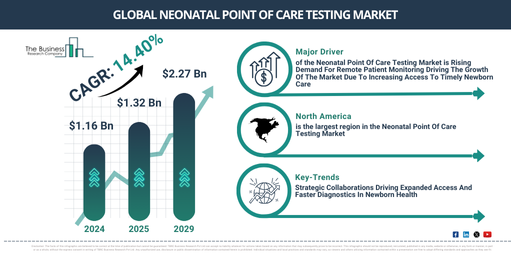
The neonatal point of care testing market has seen significant expansion in recent years. This market is projected to grow from $1.16 billion in 2024 to $1.32 billion in 2025, reflecting a compound annual growth rate (CAGR) of 14.6%. The historical growth of this market was influenced by an increasing incidence of neonatal disorders, a rising demand for swift diagnostic results, heightened awareness of neonatal health risks, a growing prevalence of preterm births, and the increasing adoption of bedside testing in hospitals.
The neonatal point of care testing market is projected to experience substantial expansion over the upcoming years. This market is anticipated to reach a valuation of $2.27 billion by 2029, demonstrating a compound annual growth rate (CAGR) of 14.4%. This anticipated growth throughout the forecast period stems from several factors, including a heightened demand for customized neonatal care, the increasing integration of portable testing devices, a stronger inclination towards early disease identification, an intensified effort to lower neonatal mortality rates, and a greater allocation of resources to sophisticated diagnostic instruments. Key developments expected during this timeframe encompass breakthroughs in biosensor technology, advancements in microfluidic diagnostic platforms, significant investment in the research and development of multiplex assays, progress in wireless neonatal monitoring systems, and novel approaches in point of care molecular testing.
Unlock Your Free Sample Report for Exclusive Market Data:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28277&type=smp
Which Drivers Are Supporting Growth Of The Neonatal Point Of Care Testing Market?
The escalating demand for remote patient monitoring is projected to propel the expansion of the neonatal point of care testing market in the coming period. Remote patient monitoring entails the delivery of healthcare services through technology, connecting patients with medical professionals from a distance. The need for remote patient monitoring is increasing due to the growing requirement for accessible and convenient medical care, particularly for individuals in remote or underserved regions. Neonatal point-of-care testing supports remote patient monitoring by providing immediate diagnostic results at the bedside, enabling healthcare providers to swiftly identify and manage crucial conditions in newborns without relying on a central laboratory. For example, in February 2023, a survey conducted by Rock Health, a US-based health technology company, among 8,014 respondents, found that 76% of people over the age of 55 had used telemedicine, and 80% of all respondents stated they had accessed care via telemedicine at some point in their lives, an increase of 8% from 72% in 2021. Therefore, the heightened demand for remote patient monitoring is anticipated to drive the growth of the neonatal point of care testing market.
What Are The Segment Types Shaping The Neonatal Point Of Care Testing Market?
The neonatal point of care testing market covered in this report is segmented as
1) By Product: Instruments, Assay
2) By Test Type: Neonatal Bilirubin Testing, Newborn Metabolic Screening, Blood Glucose Testing, Infectious Disease Screening, Electrolyte Testing, Other Test Types
3) By End-Users: Hospitals, Diagnostic Centers, Maternity And Specialty Clinics, Other End-Users
Subsegments:
1) By Instruments: Blood Gas Analyzers, Bilirubinometers, Glucose Meters, Hematology Analyzers, Multiparameter Monitors
2) By Assay: Bilirubin Assays, Glucose Assays, Infectious Disease Assays, Hematology Assays, Cardiac Marker Assays, Electrolyte Assays, Blood Gas Assays
What Trends Are Expected To Drive The Neonatal Point Of Care Testing Market?
Leading companies in the neonatal point of care testing market are prioritizing the establishment of strategic collaborations to more effectively penetrate new markets and expand their customer bases. Strategic collaborations involve alliances between organizations that merge resources and expertise to achieve common business objectives. For instance, in January 2025, QIAGEN, a Netherlands-based supplier of sample and assay technologies, entered into a partnership with Genomics England, a UK-based genomic medicine company. This collaboration has paved the way for the launch of the Generation Study, which intends to sequence the genomes of 100,000 newborns in England to identify more than 200 actionable genetic conditions. QIAGEN will furnish distinctive clinically relevant variant information for the genes included in the point-of-care sequencing test. This data will enable quicker interpretation and reporting of sequencing results, thereby supporting earlier identification and treatment of rare conditions. Officially commencing in October 2024, the study focuses on conditions that may impact roughly 3,000 newborns each year in the UK.
Which Leading Players Are Enhancing Their Neonatal Point Of Care Testing Market Presence Worldwide?
Major companies operating in the neonatal point of care testing market are Abbott Laboratories, Siemens Healthineers AG, Koninklijke Philips N.V., GE Healthcare Technologies Inc., Konica Minolta Inc., Dräger Ag Co. Kg, Bio-Rad Laboratories Inc., Masimo Corporation, QIAGEN Inc., Radiometer Ltd., Contec Medical Systems Co. Ltd., Owen Mumford Ltd., Nova Biomedical, Heal Force Bio Meditech Holdings Ltd., Edan Instruments Inc., Trivitron Healthcare, Bistos Co. Ltd., Bilimetrix S.r.I, GINEVRI srl, Hadleigh Health Technologies.
Download The Full Report For Exclusive Market Findings:
Which Region Shows The Highest Potential For Future Expansion In The Neonatal Point Of Care Testing Market?
North America was the largest region in the neonatal point of care testing market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in neonatal point of care testing report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East and Africa.
Request A Customized Version Of The Neonatal Point Of Care Testing Market Report:
https://www.thebusinessresearchcompany.com/customise?id=28277&type=smp
Browse Through More Reports Similar to the Global Neonatal Point Of Care Testing Market 2025, By The Business Research Company
Neonatal Infant Care Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/neonatal-infant-care-global-market-report
Pediatric And Neonatal Testing Kit Global Market Report 2025
Prenatal Testing And Newborn Screening Global Market Report 2025
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model