Neurology Clinical Trials Market Anticipated to Record Steady Gains, Advancing to $8.04 Billion by 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the neurology clinical trials market from 2026–2035 with trusted insights from The Business Research Company
What upper market value could the Neurology Clinical Trials Market reach by 2030 starting from 2026 levels?
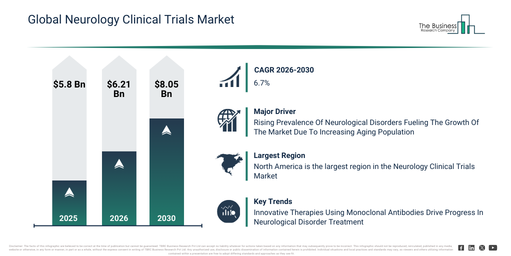
The neurology clinical trials market size has exhibited robust growth over recent years. It is projected to increase from $5.8 billion in 2025 to $6.21 billion in 2026, achieving a compound annual growth rate (CAGR) of 7.0%. This historical growth stems from factors such as an escalation in neurological disease prevalence, the broadening of pharmaceutical research pipelines, increased capital allocated to neurology drug development, the accessibility of advanced imaging tools, and a greater involvement in clinical research.
The neurology clinical trials market is projected for significant expansion over the coming years, reaching a valuation of $8.05 billion by 2030, driven by a compound annual growth rate (CAGR) of 6.7%. This anticipated growth during the forecast period is primarily fueled by several factors, including the expanding application of AI-driven trial analytics, a heightened demand for swifter trial execution, the broader adoption of patient-centric trial models, the increasing incorporation of remote monitoring technologies, and a sharper focus on uncommon neurological conditions. Key trends anticipated within this period encompass the growing acceptance of adaptive clinical trial designs, the increased deployment of digital patient monitoring tools, an escalating emphasis on biomarker-based neurology trials, the wider implementation of decentralized clinical trial models, and an intensified focus on generating real-world evidence.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25847&type=smp
Which Drivers Are Influencing Long-Term Growth In The Neurology Clinical Trials Market?
A rise in neurological disorder cases is anticipated to drive the expansion of the neurology clinical trials market in the coming years. These disorders are defined as ailments impacting the brain, spinal cord, or nerves, thereby hindering the nervous system’s regular operation. The primary cause of this increase is the aging demographic, given that seniors are more prone to age-associated illnesses like Alzheimer’s and Parkinson’s disease. Neurology clinical trials play a crucial role in addressing neurological disorders through the evaluation of novel treatments, medications, or therapeutic approaches aimed at enhancing diagnosis, alleviating symptoms, delaying disease progression, and bettering patient results. For example, data from September 2024 by the Australian Institute of Health and Welfare, an Australia-based government body, projects that the population of Australians afflicted with dementia will more than double, escalating from 411,100 in 2023 to 849,300 by 2058. Furthermore, by 2058, an estimated 315,500 men and 533,800 women are expected to be living with dementia. Consequently, the expanding occurrence of neurological conditions is fueling the growth of the neurology clinical trials market.
How Is The Neurology Clinical Trials Market Divided Into Segments?
The neurology clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV
2) By Study Design: Interventional, Observational, Expanded Access
3) By Indication: Epilepsy, Parkinson’s Disease (PD), Huntington’s Disease, Stroke, Traumatic Brain Injury (TBI), Amyotrophic Lateral Sclerosis (ALS), Multiple Sclerosis, Other Indications
4) By End-User: Hospitals, Clinics, Research Institutes, Other End- Users
Subsegments:
1) By Phase I: Drug Safety Evaluation, Pharmacokinetics Assessment, Dosage Range Finding, Tolerability Studies
2) By Phase II: Efficacy Evaluation, Dose Response Studies, Therapeutic Effectiveness, Biomarker Identification
3) By Phase III: Large Scale Efficacy Studies, Safety Monitoring, Comparative Effectiveness, Risk Benefit Analysis
4) By Phase IV: Post Marketing Surveillance, Long Term Safety Follow Up, Real World Evidence Collection, Quality Of Life Assessment
What Trends Are Driving The Growth Trajectory Of The Neurology Clinical Trials Market?
Leading companies active in the neurology clinical trials market are concentrating on developing innovative solutions, such as monoclonal antibodies, to target specific neurological pathways, enhance treatment efficacy, and accelerate the creation of new therapies. A monoclonal antibody is a laboratory-produced molecule engineered to recognize and bind to specific proteins or antigens, frequently utilized to precisely target disease mechanisms in treatments and diagnostics. For instance, in July 2024, Eli Lilly and Company, a U.S.-based pharmaceutical company, launched Kisunla (donanemab-azbt) for treating early symptomatic Alzheimer’s disease. This treatment works to slow cognitive and functional decline, enabling eligible patients more time to maintain independence and engage in daily life. It specifically targets and reduces brain amyloid plaques, with clinical trials indicating up to a 35% slowing of disease progression compared to a placebo in individuals with less advanced disease.
Which Major Firms Are Strengthening Their Position In The Neurology Clinical Trials Market?
Major companies operating in the neurology clinical trials market are AbbVie Inc., Sanofi S.A., Novartis AG, Eli Lilly and Company, Teva Pharmaceutical Industries Ltd., IQVIA Holdings Inc., Biogen Inc., ICON plc., Syneos Health, Charles River Laboratories International Inc., Parexel International Corporation, CMIC Holdings Co. Ltd., Medpace Holdings Inc., PSI CRO AG., Supernus Pharmaceuticals Inc., Worldwide Clinical Trials LLC., Ergomed plc., Biotrial S.A.S., Clinilabs Drug Development Corporation, Lindus Health Ltd.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/neurology-clinical-trials-global-market-report
Which Global Regions Are Shaping The Competitive Landscape Of The Neurology Clinical Trials Market?
North America was the largest region in the neurology clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the neurology clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Neurology Clinical Trials Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=25847&type=smp
Browse Through More Reports Similar to the Global Neurology Clinical Trials Market 2026, By The Business Research Company
Pediatric Clinical Trials Market Report 2026
https://www.thebusinessresearchcompany.com/report/pediatric-clinical-trials-global-market-report
Clinical Trial Imaging Market Report 2026
https://www.thebusinessresearchcompany.com/report/clinical-trial-imaging-global-market-report
Nephrologists Market Report 2020 2030 Covid 19 Implications And Growth 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model