Next-Generation In Vitro Diagnostics (IVD) Market Outlook Highlighting Key Drivers And Market Evolution
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Is The Estimated Market Size Of The Next-Generation In Vitro Diagnostics (IVD) Market In 2026, And How Will Its Value Evolve By 2030?_x000D_
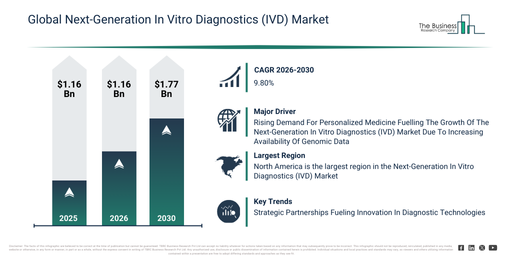
The market size for next-generation in vitro diagnostics (ivd) has experienced rapid expansion in recent years. It is projected to increase from $117.97 billion in 2025 to $129.89 billion in 2026, at a compound annual growth rate (CAGR) of 10.1%. During the preceding period, this growth was primarily fueled by the rising prevalence of infectious diseases, the expanding burden of chronic diseases, increased adoption of molecular diagnostics, the enlargement of clinical laboratory infrastructure, and government funding allocated for diagnostic programs._x000D_
_x000D_
The next-generation in vitro diagnostics (ivd) market size is anticipated to experience substantial growth in the coming years. It is predicted to expand to $188.69 billion in 2030, exhibiting a compound annual growth rate (CAGR) of 9.8%. This expansion over the forecast period is propelled by increasing demand for precision medicine, the broadening of genomic testing applications, the growing acceptance of point-of-care diagnostics, rising healthcare digitization, and heightened investments in diagnostic R&D. Prominent trends throughout the forecast period include a transition toward personalized and companion diagnostics, the rapid adoption of point-of-care and decentralized testing, the integration of multiplex and high-throughput assays, an escalating demand for early disease detection and screening, and the advancement of non-invasive and liquid biopsy diagnostics._x000D_
_x000D_
#Access Your Free Sample Report For In-Depth Market Analysis:#_x000D_
_x000D_
Which Major Drivers Are Influencing The Expansion Of The Next-Generation In Vitro Diagnostics (IVD) Market?_x000D_
The next-generation in vitro diagnostics (IVD) market is projected to expand, driven by the escalating need for personalized medicine. This healthcare strategy, known as precision medicine, involves tailoring disease prevention and treatment according to an individual’s unique genetic makeup, environmental factors, and lifestyle choices. The rise of personalized medicine stems from the increasing access to genomic data, empowering healthcare professionals to develop specific treatments aligned with a person’s distinct genetic profile, thereby improving treatment effectiveness. Next-generation in vitro diagnostics (IVD) plays a crucial role in personalized medicine by offering accurate molecular and genetic information, which facilitates the creation of customized treatment and prevention plans for patients. An example highlighting this trend is from February 2024, when the Personalized Medicine Coalition, a US-based hospital and health care organization, reported that the FDA approved 16 new personalized treatments for rare disease patients in 2023, a significant increase from six approvals in 2022. Consequently, the growing requirement for personalized medicine is fueling the expansion of the next-generation in vitro diagnostics (IVD) market._x000D_
_x000D_
How Is The Next-Generation In Vitro Diagnostics (IVD) Market Categorized Across Its Segment Groups?_x000D_
The next-generation in vitro diagnostics (ivd) market covered in this report is segmented – _x000D_
_x000D_
1) By Product Type: Consumables, Instruments, Software_x000D_
2) By Diagnostic Modality: Core Laboratory Diagnostics, Point-Of-Care Testing, Molecular Diagnostics_x000D_
3) By Application: Oncology Or Cancer, Infectious Diseases, Diabetes, Cardiology , Other Applications_x000D_
4) By End-User: Hospitals And Clinics, Diagnostic Laboratories, Academic And Research Institutions, Other End Users_x000D_
_x000D_
Subsegments:_x000D_
1) By Consumables: Assay Kits And Reagents, Specimen Bags and Tubes, Seals and Caps_x000D_
2) By Instruments: Automated Sample Preparation Systems, High-Throughput Molecular Diagnostic Analyzers, Next-Generation Sequencing Platforms, Integrated Diagnostic Workstations And Analysis Platforms, Digital PCR Systems And Advanced Real-Time PCR Instruments_x000D_
3) By Software: Laboratory Information Management Systems, Specimen Tracking Software Modules, Cloud-Based Tracking Software_x000D_
_x000D_
Which Trends Are Shaping The Next-Generation In Vitro Diagnostics (IVD) Market?_x000D_
Prominent companies active within the next-generation in vitro diagnostics (IVD) sector are implementing a strategic partnerships strategy. This method, applied to next-generation in vitro diagnostics, encompasses working alongside technology firms, research bodies, and healthcare providers to speed up innovation, improve diagnostic precision, and broaden the availability of advanced testing options for individuals, enterprises, and governmental entities. These strategic collaborations describe a system where organizations capitalize on one another’s strengths and assets to attain collective advantages and successful outcomes. As an illustration, in October 2024, Yourgene Health, a biotechnology company headquartered in the UK, collaborated with Genetix, a human genetic diagnosis company based in Colombia, to introduce Colombia’s inaugural local non-invasive prenatal testing (NIPT) service. This undertaking signifies a substantial progression in regional prenatal care, providing prospective parents with quicker, more dependable, and economically viable access to sophisticated genetic screening. The service employs Yourgene Health’s IONA Nx NIPT Workflow, a complete and CE-marked in vitro diagnostic (IVD) solution. Being highly adaptable and scalable, this workflow can accommodate diverse sample volumes, empowering Genetix to deliver a quality-assured prenatal screening service throughout Colombia._x000D_
_x000D_
Which Firms Are Influencing Competition In The Next-Generation In Vitro Diagnostics (IVD) Market?_x000D_
Major companies operating in the next-generation in vitro diagnostics (ivd) market are F Hoffmann La Roche AG, Thermo Fisher Scientific Inc, Abbott Laboratories, Becton Dickinson And Company, Agilent Technologies Inc, Illumina Inc, Sysmex Corporation, Bio Rad Laboratories Inc, Revvity Inc, Exact Sciences Corporation, QIAGEN NV, DiaSorin SpA, Singular Genomics Systems Inc, Guardant Health Inc, Pacific Biosciences Of California Inc, Transasia Bio Medicals Ltd, J Mitra And Co Pvt Ltd, Roche Diagnostics International AG, Hologic Inc, Genetron Health Inc, Seegene Inc, BGI Genomics Co Ltd, Oxford Nanopore Technologies plc _x000D_
_x000D_
#Get The Full Next-Generation In Vitro Diagnostics (IVD) Market Report:#_x000D_
_x000D_
Which Region Leads The Next-Generation In Vitro Diagnostics (IVD) Market In Terms Of Market Share?_x000D_
North America was the largest region in the next-generation in vitro diagnostics (IVD) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the next-generation in vitro diagnostics (ivd) market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa._x000D_
_x000D_
#Request A Customized Next-Generation In Vitro Diagnostics (IVD) Market Report For Competitive Insights:#_x000D_
_x000D_
#Browse Through More Reports Similar to the Global Next-Generation In Vitro Diagnostics (IVD) Market 2026, By The Business Research Company#_x000D_
_x000D_
In Vitro Diagnostics Market Report 2026_x000D_
https://www.thebusinessresearchcompany.com/report/in-vitro-diagnostics-global-market-report_x000D_
_x000D_
In Vitro Diagnostic Ivd In Cardiology And Neurology Market Report 2026_x000D_
_x000D_
Bioinformatics In In Vitro Diagnostics Ivd Testing Market Report 2026_x000D_
_x000D_
#Get in touch with us:#_x000D_
_x000D_
The Business Research Company: https://www.thebusinessresearchcompany.com/_x000D_
Americas +1 310-496-7795_x000D_
Asia +44 7882 955267 & +91 8897263534_x000D_
Europe +44 7882 955267_x000D_
Email us at info@tbrc.info_x000D_
_x000D_
#Follow us on:#_x000D_
_x000D_
LinkedIn: https://in.linkedin.com/company/the-business-research-company_x000D_
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ_x000D_
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model
[KClientError] [REQ_ERR: OPERATION_TIMEDOUT] [KTrafficClient] Something is wrong.