Nuclear Protein In Testis Midline Carcinoma Treatment Sector Forecast Shows Robust Momentum, Climbing to $42.2 Billion With 13.6% CAGR
Uncover key drivers, emerging technologies, and competitive movements shaping the nuclear protein in testis midline carcinoma treatment market from 2026–2035 with trusted insights from The Business Research Company
What upper market value could the Nuclear Protein In Testis Midline Carcinoma Treatment Market reach by 2030 starting from 2026 levels?
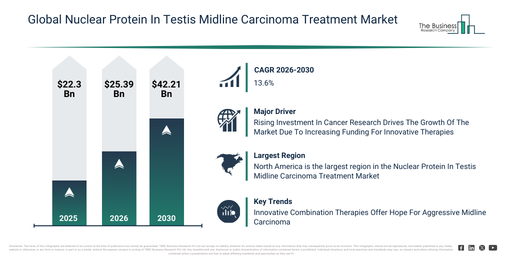
The nuclear protein in testis midline carcinoma treatment market has experienced rapid expansion in recent years. It is projected to grow from $22.3 billion in 2025 to $25.39 billion in 2026, at a compound annual growth rate (CAGR) of 13.8%. This historical growth can be attributed to several factors, including improved recognition of nut midline carcinoma, advancements in molecular diagnostic capabilities, the availability of combination chemotherapy protocols, the expansion of specialized oncology treatment centers, and increased rare cancer awareness.
The nuclear protein in testis midline carcinoma treatment market is projected to experience substantial expansion over the upcoming years. This market is anticipated to reach $42.21 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 13.6%. Factors contributing to this growth during the forecast period include greater financial commitments to targeted oncology drug development, a rise in the use of precision oncology strategies, broader immunotherapy research efforts for uncommon cancers, intensified partnerships between academic institutions and pharmaceutical firms, and enhanced regulatory backing for orphan drugs. Key developments expected within this period encompass the increased creation of targeted nut fusion inhibitors, an uptick in molecular profiling for guiding treatment choices, wider integration of personalized therapy plans, an increase in clinical trials for rare cancers, and a stronger emphasis on combined treatment methodologies.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=30339&type=smp
Which Drivers Are Influencing Long-Term Growth In The Nuclear Protein In Testis Midline Carcinoma Treatment Market?
The expanding financial commitment to cancer research is anticipated to boost the nuclear protein in testis midline carcinoma treatment market in the coming years. This investment involves governments, organizations, and private entities dedicating funds to studies, clinical trials, and new developments focused on understanding, preventing, and treating cancer. The growth in cancer research investment stems from the rising occurrence of cancer, which necessitates enhanced treatments and novel therapies. Such increased funding aids nuclear protein in testis (NUT) midline carcinoma treatment by facilitating the creation of targeted therapies, sophisticated diagnostics, and advanced treatment methods. For example, in September 2024, Civil Society Media Limited, a UK-based media company, reported that Cancer Research UK allocated $0.0001737 billion to fundraising efforts in 2023–2024, representing an increase of $0.0305 billion compared to the $0.136 billion spent the previous year. Consequently, greater investment in cancer research is fueling the expansion of the nuclear protein in testis midline carcinoma treatment market. The rising volume of clinical trials is anticipated to stimulate expansion in the nuclear protein in testis midline carcinoma treatment market in the future. Clinical trials are meticulously planned research studies involving human participants, designed to assess the safety, effectiveness, and utility of new medical therapies, medications, or interventions. The rise in clinical trials is attributed to the growing incidence of various diseases, which generates an urgent demand for novel and efficient treatments. These trials contribute to advancing nuclear protein in testis (NUT) midline carcinoma treatment by scrutinizing the safety, effectiveness, and optimal application of new therapies, thereby expediting the development of precise and impactful treatment options for patients. For example, in December 2024, the Association of the British Pharmaceutical Industry, a UK-based trade association, reported that the total number of industry clinical trials launched in the UK reached 426 in 2023, an increase from 411 trials in 2022, marking a second consecutive year of growth. Consequently, the growing number of clinical trials is propelling the nuclear protein in testis midline carcinoma treatment market forward.
How Is The Nuclear Protein In Testis Midline Carcinoma Treatment Market Divided Into Segments?
The nuclear protein in testis midline carcinoma treatment market covered in this report is segmented –
1) By Treatment: Chemotherapy, Targeted Therapy, Immunotherapy, Radiation Therapy, Other Treatments
2) By Technology: Molecular Profiling, Personalized Medicine Approaches, Next-Generation Sequencing (NGS), Other Technologies
3) By Route Of Administration: Oral, Intravenous, Other Routes Of Administration
4) By Distribution Channels: Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Direct Sales
5) By End-Use: Hospitals, Specialty Cancer Clinics, Ambulatory Surgical Centers, Cancer Research Institutes And Academic Hospitals, Other End-Uses
Subsegments:
1) By Chemotherapy: Platinum-Based Chemotherapy, Combination Chemotherapy, Single-Agent Chemotherapy, Adjuvant Chemotherapy
2) By Targeted Therapy: Tyrosine Kinase Inhibitors, Small Molecule Inhibitors, Monoclonal Antibodies, Epigenetic Modulators
3) By Immunotherapy: Immune Checkpoint Inhibitors, Adoptive Cell Therapy, Cancer Vaccines, Cytokine Therapy
4) By Radiation Therapy: External Beam Radiation, Stereotactic Radiation, Proton Therapy, Brachytherapy
5) By Other Treatments: Surgical Resection, Supportive Care, Palliative Care, Experimental Therapies
How Are New Market Trends Shaping The Landscape Of The Nuclear Protein In Testis Midline Carcinoma Treatment Market?
Leading companies within the nuclear protein in testis midline carcinoma treatment market are prioritizing innovative therapeutic strategies, particularly combination therapies, to enhance treatment efficacy and improve patient outcomes. Combination therapies are defined as treatments that integrate two or more distinct drugs or approaches to bolster effectiveness and lessen resistance against a disease. As an illustration, in July 2025, Zenith Epigenetics Ltd., a Canada-based clinical-stage biotechnology company, obtained U.S. Food and Drug Administration (FDA) Fast Track designation for ZEN 3694 when combined with abemaciclib. This combination is intended for treating patients with metastatic or unresectable NUT carcinoma who have completed at least one prior chemotherapy regimen. The objective of this designation is to accelerate the development and regulatory review of treatments for serious conditions with unaddressed medical needs. These therapies, which include an oral BET inhibitor, target the oncogenic NUTM1 fusion protein driving NUT carcinoma, while abemaciclib, a CDK4/6 inhibitor, aids in inhibiting cancer cell proliferation.
Which Major Industry Participants Are Leading The Nuclear Protein In Testis Midline Carcinoma Treatment Market Growth?
Major companies operating in the nuclear protein in testis midline carcinoma treatment market are Johnson And Johnson, F. Hoffmann-La Roche Limited, Merck & Company Inc., Pfizer Inc., AbbVie Incorporated, Novartis International AG, Bristol-Myers Squibb Company, GlaxoSmithKline Public Limited Company, Takeda Pharmaceutical Company Limited, Gilead Sciences Inc., Siemens Healthineers AG, Biogen Inc., Illumina Inc., Varian Medical Systems Inc., Elekta AB, Accuray Incorporated, ViewRay Inc., Arcus Biosciences Inc., Xilio Therapeutics Inc., Zenith Epigenetics Inc.
Access The Complete Report For Deeper Market Insights:
Which Geographic Areas Are Emerging As Strong Markets For The Nuclear Protein In Testis Midline Carcinoma Treatment Market?
North America was the largest region in the nuclear protein in testis midline carcinoma treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the nuclear protein in testis midline carcinoma treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Nuclear Protein In Testis Midline Carcinoma Treatment Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=30339&type=smp
Browse Through More Reports Similar to the Global Nuclear Protein In Testis Midline Carcinoma Treatment Market 2026, By The Business Research Company
Hereditary Cancer Testing Market Report 2026
https://www.thebusinessresearchcompany.com/report/hereditary-cancer-testing-global-market-report
Prostate Cancer Nuclear Medicine Diagnostics Market Report 2026
Nuclear Medicine Market Report 2026
https://www.thebusinessresearchcompany.com/report/nuclear-medicine-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model