Oncology Clinical Trials Market Trends and Insights: Global Forecast to 2030
Uncover key drivers, emerging technologies, and competitive movements shaping the oncology clinical trials market from 2026–2035 with trusted insights from The Business Research Company
What is the expected shift in the Oncology Clinical Trials Market’s size between 2026 and 2030?
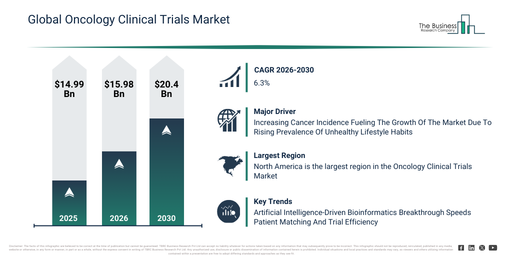
The oncology clinical trials market size has demonstrated robust growth recently. It is forecast to expand from $14.99 billion in 2025 to $15.98 billion in 2026, achieving a compound annual growth rate (CAGR) of 6.6%. This past expansion is attributable to factors such as limited therapeutic options for cancer patients, a global increase in cancer cases, enhanced R&D investments by pharmaceutical and biotech firms, a growing number of academic clinical research facilities, and heightened recognition of the importance of clinical trials.
The oncology clinical trials market is projected to experience robust expansion in the coming years. This market is anticipated to reach a valuation of $20.4 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 6.3%. Factors contributing to this growth over the forecast period encompass the creation of individualized oncology treatments, the proliferation of multicenter and international studies, the implementation of digital tools for patient monitoring, the incorporation of AI and big data into trial formulation, and enhanced government and private financial support for oncology investigations. Significant developments projected for the forecast timeframe involve the broadening of immunotherapy and targeted therapy clinical trials, a heightened emphasis on multicenter and randomized controlled studies, greater funding for post-marketing surveillance and long-term safety analyses, the increasing uptake of cell and gene therapy trials, and the incorporation of biomarker and pharmacogenomics research into oncology studies.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=27371&type=smp
Which Drivers Are Shaping Strategic Decisions In The Oncology Clinical Trials Market?
The rising occurrence of cancer is projected to fuel the expansion of the oncology clinical trials market in the future. Cancer incidence is defined as the count of new cancer cases identified within a particular demographic over a set duration. This growing incidence of cancer is mainly attributable to detrimental lifestyle choices, such as inadequate nutrition, lack of physical activity, tobacco use, and excessive alcohol consumption, which heighten the likelihood of developing diverse forms of cancer. Oncology clinical trials contribute to the progress of cancer treatment by assessing novel therapies and interventions, thereby enhancing patient results via research grounded in evidence. These trials also quicken medical breakthroughs by examining safety and effectiveness, thus propelling advancements in the prevention, diagnosis, and care of cancer. As an illustration, in October 2025, the National Health Service (NHS), a government health agency based in the UK, reported 354,820 new cancer diagnoses in 2023, averaging 972 daily. This figure represents an increase of 8,605 compared to 2022. Consequently, the escalating number of cancer cases is a key factor boosting the expansion of oncology clinical trials.
What Segment Categories Are Covered In The Oncology Clinical Trials Market?
The oncology clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV
2) By Study Design: Interventional, Observational, Expanded Access
3) By Therapeutic Modality: Immunotherapy, Targeted Therapy, Chemotherapy, Cell And Gene Therapy, Other Therapeutic Modalities
4) By Indication: Lung Cancer, Breast Cancer, Colorectal Cancer, Prostate Cancer, Blood Cancer, Other Indications
5) By Sponsor Type: Pharmaceutical And Biotech Companies, Academic And Research Institutes, Government Organizations, Other Sponsor Types
Subsegments:
1) By Phase I: Safety And Dosage Studies, Pharmacokinetics And Pharmacodynamics Studies, Biomarker Identification Studies
2) By Phase II: Efficacy And Side Effects Studies, Dose-Response Relationship Studies, Therapeutic Effectiveness Studies
3) By Phase III: Large-Scale Efficacy Studies, Comparative Effectiveness Studies, Multicenter Randomized Controlled Trials
4) By Phase IV: Post Marketing Surveillance Studies, Long-Term Safety Studies, Quality Of Life Studies
What Upcoming Trends Are Likely To Define The Future Path Of The Oncology Clinical Trials Market?
Major companies operating in the oncology clinical trials market are concentrating on creating sophisticated technological solutions, such as advanced bioinformatics platforms, to improve data analysis, hasten patient enrollment, and optimize trial results. Advanced bioinformatics platforms are intricate software systems that employ computational tools and algorithms to gather, analyze, and interpret complex biological and clinical information for research and healthcare applications. For instance, in September 2023, Deep 6 AI, a US-based healthcare technology firm, introduced an AI-driven genomics module designed to expedite enrollment in precision medicine and oncology clinical trials. This module utilizes artificial intelligence and natural language processing to extract both structured and unstructured genomics data from electronic medical records, allowing for the real-time identification and precise matching of patients with specific genetic markers. It empowers researchers to explore millions of records for over 19,000 genes and 30,000 mutation names, significantly speeding up patient recruitment and enhancing the effectiveness of clinical trial design.
Which Major Firms Are Strengthening Their Position In The Oncology Clinical Trials Market?
Major companies operating in the oncology clinical trials market are Pfizer Inc., Johnson & Johnson, Merck And Co. Inc., AbbVie Inc., Bayer AG, Novartis International AG, Sanofi S.A., Bristol-Myers Squibb Company, AstraZeneca plc, GSK plc, Takeda Pharmaceutical Company Limited, Eli Lilly and Company, Gilead Sciences Inc., Amgen Inc., Boehringer Ingelheim GmbH, Regeneron Pharmaceuticals Inc., Genentech Inc., BeiGene Ltd., Seagen Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/oncology-clinical-trials-global-market-report
Which Region Shows The Strongest Potential For Future Expansion In The Oncology Clinical Trials Market?
North America was the largest region in the oncology clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the oncology clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Oncology Clinical Trials Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=27371&type=smp
Browse Through More Reports Similar to the Global Oncology Clinical Trials Market 2026, By The Business Research Company
Concrete Materials Market Report 2026
https://www.thebusinessresearchcompany.com/report/concrete-materials-global-market-report
Decorative Concrete Market Report 2026
https://www.thebusinessresearchcompany.com/report/decorative-concrete-global-market-report
Cellular Concrete Market Report 2026
https://www.thebusinessresearchcompany.com/report/cellular-concrete-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model