Orphan Drugs Service Sector Forecast Shows Robust Momentum, Climbing to $93.93 Billion With 16.6% CAGR
Uncover key drivers, emerging technologies, and competitive movements shaping the orphan drugs service market from 2026–2035 with trusted insights from The Business Research Company
What level of CAGR-driven expansion is anticipated in the Orphan Drugs Service Market between 2026 and 2030?
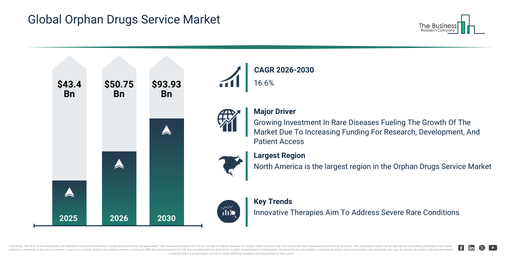
The orphan drugs service market has experienced rapid expansion in recent years. Its valuation is projected to increase from $43.4 billion in 2025 to $50.75 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 16.9%. Historically, this growth has been driven by factors such as growth in rare disease identification, expansion of orphan drug legislation, increasing investment in rare disease research, availability of regulatory incentives, and the rising role of patient advocacy groups.
The orphan drugs service market is projected for swift expansion in the upcoming years. Its value is anticipated to reach $93.93 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 16.6%. This expansion during the forecast period is largely due to a heightened demand for tailored rare disease treatments, the increasing integration of cutting-edge clinical trial technologies, the widening scope of global orphan drug development pipelines, the expanded application of real-world evidence, and ongoing regulatory backing for orphan drug creation. Key trends expected within this period encompass a greater need for specialized regulatory and developmental assistance, a stronger emphasis on expedited approval routes, a surge in the utilization of data-powered rare disease research platforms, the proliferation of patient-focused clinical development frameworks, and strengthened partnerships among involved parties.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28287&type=smp
What Drivers Are Affecting Demand-Supply Dynamics In The Orphan Drugs Service Market?
The orphan drugs service market is projected to experience growth, propelled by the increasing investment in rare diseases. A rare disease is defined as a medical condition impacting a small fraction of the population, typically fewer than 1 in 2,000 people, often characterized as chronic, progressive, and potentially life-threatening or disabling. This investment is mainly driven by an increasing awareness of unaddressed medical requirements and the potential for substantial returns via regulatory incentives like orphan drug designation, market exclusivity, and decreased development expenses. Enhanced investment in rare diseases improves orphan drug services by securing more funding for research and development, facilitating the creation of treatments for previously neglected conditions. It also speeds up drug availability through support for clinical trials, regulatory approvals, and patient access, thereby expanding therapeutic options for individuals with such conditions. For example, Global Genes, a US-based nonprofit organization, reported in April 2024 that companies focused on rare disease drug development secured $7.1 billion in the first quarter of 2024, which is a 307% increase from the $1.8 billion raised during the same period in 2023. Consequently, the expanding investment in rare diseases is a key factor driving the growth of the orphan drugs service market.
What Segments Are Included Within The Orphan Drugs Service Market?
The orphan drugs service market covered in this report is segmented –
1) By Type: Congenital Disease, Genetic Disease, Tumors And Cancer, Chronic, Other Types
2) By Therapeutic Area: Oncology, Metabolic Disorders, Neurological Disorders, Hematology, Infectious Diseases, Other Rare Diseases
3) By Application: Aldult, Children
Subsegments:
1) By Congenital Disease: Heart Defects, Cleft Lip And Palate, Spina Bifida, Down Syndrome
2) By Genetic Disease: Cystic Fibrosis, Huntington Disease, Sickle Cell Anemia, Muscular Dystrophy
3) By Tumors And Cancer: Leukemia, Lymphoma, Neuroblastoma, Sarcoma
4) By Chronic: Rare Metabolic Disorders, Rare Endocrine Disorders, Rare Cardiovascular Disorders, Rare Neurological Disorders
5) By Other Types: Rare Infectious Diseases, Rare Immunological Disorders, Rare Dermatological Conditions, Rare Ophthalmological Disorders
What Trends Are Reshaping The Dynamics Of The Orphan Drugs Service Market?
Leading companies in the orphan drugs service market are concentrating on securing regulatory approvals to accelerate the development and availability of treatments for rare diseases. Regulatory approvals are official permissions from government agencies that enable a drug or treatment to be legally tested, manufactured, and marketed for patient use. For example, in February 2023, Askbio GmbH, a US-based gene therapy company, obtained the European Commission orphan drug designation for AB-1003, an investigational gene therapy for limb-girdle muscular dystrophy. This adeno-associated virus (AAV)-based therapy is designed to restore FKRP enzyme activity in muscle cells through a one-time intravenous infusion. AB-1003 has received orphan drug designation from both the European Commission and the U.S. Food and Drug Administration (FDA), alongside rare pediatric disease and fast track designations from the FDA, emphasizing the urgent unmet medical need. It is presently in a Phase 1/2 clinical trial (LION-CS101), evaluating its safety and efficacy in adults with genetically confirmed limb-girdle muscular dystrophy type 2I/R9 (LGMD2I/R9).
Who Are The Major Companies Operating In The Orphan Drugs Service Market?
Major companies operating in the orphan drugs service market are Johnson & Johnson Services Inc., F. Hoffmann-La Roche Ltd., Pfizer Inc., AbbVie Inc., Bayer AG, Sanofi S.A., Novartis AG, AstraZeneca PLC, The Bristol-Myers Squibb Company, GSK plc., Eli Lilly and Company, Takeda Pharmaceutical Company Limited, Amgen Inc., IQVIA Inc., CSL Behring GmbH, Lonza Group AG, BioMarin Pharmaceutical Inc., Fortrea Holdings Inc., Medpace Inc., Sarepta Therapeutics, Caidya Inc., Cevidra Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/orphan-drugs-service-global-market-report
Which Region Is Anticipated To See The Fastest Growth In The Orphan Drugs Service Market?
North America was the largest region in the orphan drugs service market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the orphan drugs service market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Orphan Drugs Service Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=28287&type=smp
Browse Through More Reports Similar to the Global Orphan Drugs Service Market 2026, By The Business Research Company
Drug Discovery Services Market Report 2026
https://www.thebusinessresearchcompany.com/report/drug-discovery-services-global-market-report
Child And Youth Services Market Report 2026
https://www.thebusinessresearchcompany.com/report/child-and-youth-services-global-market-report
Pharmaceuticals Market 2026
https://www.thebusinessresearchcompany.com/report/pharmaceuticals-market
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model