Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Report 2026: Emerging Trends and Growth Scenarios
Uncover key drivers, emerging technologies, and competitive movements shaping the paroxysmal nocturnal hemoglobinuria (pnh) treatment market from 2026–2035 with trusted insights from The Business Research Company
What is the estimated market size of the Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market by the end of 2030?
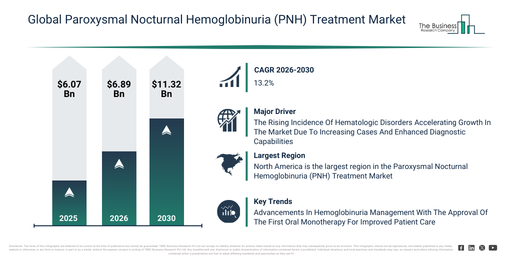
The market size for paroxysmal nocturnal hemoglobinuria (pnh) treatments has witnessed rapid growth in recent years. It is projected to expand from $6.07 billion in 2025 to $6.89 billion in 2026, achieving a compound annual growth rate (CAGR) of 13.4%. The expansion observed in the historic period can be attributed to several factors, including the restricted availability of effective targeted therapies, a dependence on supportive care and transfusions, challenges with delayed diagnosis due to the disease’s rarity, the significant risk of life-threatening thrombosis, and a lack of standardized long-term treatment protocols.
The paroxysmal nocturnal hemoglobinuria (pnh) treatment market size is set for significant expansion in the coming years, projected to reach $11.32 billion by 2030, driven by a compound annual growth rate (CAGR) of 13.2%. This anticipated growth during the forecast period can be attributed to the advancement of next-generation complement inhibitors, an expanding clinical development pipeline for oral and subcutaneous therapies, an increasing emphasis on drug innovation for orphan diseases, improved diagnosis rates through advanced testing, and better reimbursement coverage for rare hematologic disorders. Major trends identified for this period include a rise in the adoption of complement C5 inhibitor treatments, a move towards long-acting and subcutaneous forms of therapy, the development of alternative pathway inhibitors, the expansion of combination and dual complement inhibition strategies, and enhanced management of thrombotic and bone marrow complications.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=23469&type=smp
What Drivers Are Shaping Future Opportunities In The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
The growth of the paroxysmal nocturnal hemoglobinuria (PNH) treatment market is anticipated to be significantly propelled by the increasing incidence of hematologic disorders. These medical conditions encompass abnormalities affecting blood and blood-forming organs, such as red blood cells, white blood cells, platelets, and bone marrow, potentially leading to issues like anemia, clotting disorders, or malignancies. The rise in these disorders is influenced by factors like aging populations and genetic predispositions, with genetic mutations playing a crucial role in triggering abnormal blood cell production. Paroxysmal nocturnal hemoglobinuria (PNH) treatment offers a positive approach to managing hematologic disorders, providing targeted therapies that alleviate complement-mediated hemolysis, diminish thrombotic complications, and improve patient survival rates. As an illustration, Cancer Research UK stated in July 2024 that the average number of new leukemia cases in the UK is expected to increase from approximately 12,400 in 2023-2025 to about 14,500 by 2038-2040. This escalating prevalence of hematologic disorders is thus a primary driver for the expansion of the paroxysmal nocturnal hemoglobinuria (PNH) treatment market.
Which Segment Groups Are Influencing The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
The paroxysmal nocturnal hemoglobinuria (pnh) treatment market covered in this report is segmented –
1) By Treatment Type: Complement C5 Inhibitors, Anti-Complement Component 5a Receptor (C5aR) Monoclonal Antibodies, Alternative Pathway Inhibitors, Hematopoietic Stem Cell Transplantation, Other Therapies
2) By Route Of Administration: Intravenous, Subcutaneous, Oral
3) By Market Availability: Approved, In Pipeline, Investigational
4) By End-Use: Hospitals, Specialty Clinics, Homecare Settings, Other End-Users
Subsegments:
1) By Complement: C5 InhibitorsEculizumab (Soliris), Ravulizumab (Ultomiris), Emerging C5 Inhibitors, Biosimilars Of C5 Inhibitors,
2) By Anti-Complement Component 5a Receptor Monoclonal Antibodies: Avacopan (Tavneos), Other Investigational C5aR mAbs, Monotherapy vs Combination Therapy, Clinical Trial Phase (Phase I, II, III)
3) By Alternative Pathway Inhibitors: C3 Inhibitors, Factor B Inhibitors, Factor D Inhibitors, Targeting Complement Amplification, Combination Therapies And Dual Inhibition
4) By Hematopoietic Stem Cell Transplantation (HSCT): Autologous HSCT vs. Allogeneic HSCT, Pre-Transplant Conditioning Therapies, Post-Transplant Immunosuppressive Therapies
5) By Other Therapies: Immunosuppressive Therapy, Supportive Care, Anticoagulation Therapy, Gene Therapy
Which Trends Are Guiding The Evolution Of The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market?
Leading companies in the paroxysmal nocturnal hemoglobinuria (PNH) treatment market are concentrating on developing innovative solutions, such as oral factor B inhibitors, to introduce a new treatment option that improves therapeutic effectiveness and enhances patient convenience in managing PNH. An oral Factor B inhibitor is a targeted small-molecule drug designed to selectively inhibit Factor B in the alternative complement pathway, aiming to regulate immune system overactivation and alleviate inflammation in complement-mediated diseases. For instance, in December 2023, Novartis AG, a Switzerland-based pharmaceutical corporation, announced the Food and Drug Administration (FDA) approval of Fabhalta (iptacopan) as the first oral monotherapy for the treatment of paroxysmal nocturnal hemoglobinuria (PNH). This novel Factor B inhibitor offers a more comprehensive strategy for controlling complement-mediated hemolysis by targeting the alternative complement pathway at an earlier stage, leading to improved hemoglobin levels and a reduced requirement for blood transfusions. The approval of Fabhalta represents a groundbreaking advancement in PNH treatment by providing enhanced efficacy, better patient compliance, and an alternative to intravenous therapies, thus transforming the treatment landscape for this rare hematologic disorder.
Who Are The Prominent Global Companies Shaping The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Landscape?
Major companies operating in the paroxysmal nocturnal hemoglobinuria (pnh) treatment market are F. Hoffmann-La Roche Ltd, Novartis AG, Mayo Clinic, Cleveland Clinic Foundation, Johns Hopkins Medicine, Memorial Sloan Kettering Cancer Center, Stanford Health Care, University of California San Francisco (UCSF) Health, Emory Healthcare, Cedars-Sinai Medical Center, Duke University Health System, Mount Sinai Health System, Dana-Farber Cancer Institute, Swedish Orphan Biovitrum, Alnylam Pharmaceuticals, BioCryst Pharmaceuticals, Apellis Pharmaceuticals, Omeros Corporation, Takeda Pharmaceutical Company, Sanofi S A, Pfizer Inc, Akari Therapeutics Plc, NovelMed Therapeutics, Ionis Pharmaceuticals, Johnson & Johnson, Biogen Inc.
Access The Complete Report For Deeper Market Insights:
How Does The Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Perform Across Major Global Regions?
North America was the largest region in the paroxysmal nocturnal hemoglobinuria (PNH) treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the paroxysmal nocturnal hemoglobinuria (pnh) treatment market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=23469&type=smp
Browse Through More Reports Similar to the Global Paroxysmal Nocturnal Hemoglobinuria (PNH) Treatment Market 2026, By The Business Research Company
Acquired Hemophilia Treatment Market Report 2026
https://www.thebusinessresearchcompany.com/report/acquired-hemophilia-treatment-global-market-report
Hemoglobinopathies Treatment Market Report 2026
https://www.thebusinessresearchcompany.com/report/hemoglobinopathies-treatment-global-market-report
Hemoglobin Testing Market Report 2026
https://www.thebusinessresearchcompany.com/report/hemoglobin-testing-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model