PD-L1 And PIK3CA Testing Market Growth Opportunities: Insights from 2025–2029
Unlock Global Insights – Save 20% on Market Reports with Code ONLINE20
What Is The Anticipated Size Of The PD-L1 And PIK3CA Testing Market Over The Five-Year Period 2025–2029?
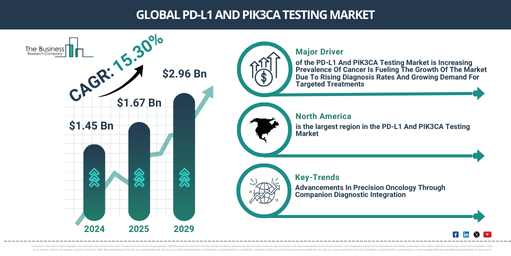
The PD-L1 and PIK3CA testing market size has experienced considerable expansion in recent years. It is projected to expand from $1.45 billion in 2024 to $1.67 billion in 2025, at a robust compound annual growth rate (CAGR) of 15.6%. The historical growth witnessed can be ascribed to several factors, including a rise in clinical trials for regenerative medicine, an increasing focus on personalized medicine, the expansion of academic and industry collaborations, enhanced awareness of cell therapy efficacy and safety, and the rapid progression and initial approvals of CAR-T and other autologous cell therapies.
The PD-L1 and PIK3CA testing market is projected to experience substantial growth over the next few years. It is forecast to expand to $2.96 billion by 2029, achieving a compound annual growth rate (CAGR) of 15.3%. This anticipated growth during the forecast period can be linked to the increasing adoption of personalized and precision medicine, an expanding pipeline of cell and gene therapies, a rise in clinical trials and commercial approvals, growing investment in regenerative medicine, and the rising incidence of chronic and rare diseases. Prominent trends for the upcoming period include the advancement of bioinformatics in cellular research, the integration of cell therapy tracking with digital health platforms, enhanced artificial intelligence (AI) and machine learning integration, innovation in cloud-based data management systems, and the incorporation of wearable biosensors.
Unlock Your Free Sample Report for Exclusive Market Data:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28289&type=smp
What Are The Key Drivers Of Growth In The PD-L1 And PIK3CA Testing Market?
The rising occurrence of cancer is projected to drive expansion in the PD-L1 and PIK3CA testing market in the future. Cancer, a condition marked by the unchecked proliferation and dissemination of atypical cells, can infiltrate adjacent tissues and potentially spread to distant areas. This heightened cancer prevalence stems from escalating lifestyle-associated risks and an aging global populace, resulting in elevated diagnosis rates and an increased need for timely detection and potent therapeutic approaches. PD-L1 and PIK3CA testing facilitates cancer management through precise diagnosis and the direction of targeted therapies, enabling personalized treatment plans and enhanced patient results. For example, data from September 2024, provided by the American Cancer Society, a US-based non-profit organization, indicates that worldwide male cancer cases are anticipated to climb by 84% by 2029, moving from 10.3 million in 2022 to 19 million. Consequently, the escalating incidence of cancer is fueling the expansion of the PD-L1 and PIK3CA testing market.
What Are The Segment Types Shaping The PD-L1 And PIK3CA Testing Market?
The pd-l1 and pik3ca testing market covered in this report is segmented as
1) By Test Type: Immunohistochemistry, Next-Generation Sequencing, Polymerase Chain Reaction, Other Test Types
2) By Sample Type: Tissue Samples, Liquid Biopsy Samples
3) By Distribution Channel: Direct Sales, Distributors And Resellers, Online Sales Platforms
4) By Applications: Cancer Diagnosis, Drug Development, Research, Other Applications
5) By End User: Hospitals And Clinics, Laboratories, Pharmaceutical Companies
Subsegments:
1) By Immunohistochemistry: Tissue Staining, Protein Expression Analysis, Tumor Microenvironment Assessment
2) By Next-Generation Sequencing: Whole Genome Sequencing, Targeted Gene Sequencing, Exome Sequencing, Ribonucleic Acid (RNA) Sequencing
3) By Polymerase Chain Reaction: Quantitative Polymerase Chain Reaction, Reverse Transcription Polymerase Chain Reaction, Digital Polymerase Chain Reaction
4) By Other Test Types: In Situ Hybridization (ISH), Sanger Sequencing, Flow Cytometry, Microarray-Based Assays
What Long-Term Trends Will Shape The Future Growth Of The PD-L1 And PIK3CA Testing Industry?
Companies operating within the PD-L1 and PIK3CA testing market are concentrating on technological enhancements, such as integrating companion diagnostics, to refine therapeutic targeting, boost treatment effectiveness, and support individualized medicine approaches in oncology. Companion diagnostic integration refers to advanced, technology-driven testing systems developed concurrently with targeted medications to identify patient demographics most likely to benefit from specific treatments based on their biomarker profiles. As an illustration, in October 2024, F. Hoffmann-La Roche AG, a Switzerland-based biotechnology and pharmaceutical company, introduced Itovebi (inavolisib) in combination with palbociclib and fulvestrant for adults diagnosed with endocrine-resistant, PIK3CA-mutated, HR-positive, HER2-negative advanced or metastatic breast cancer. The Food and Drug Administration (FDA), a US-based federal agency, granted approval for this regimen, based on the pivotal Phase III INAVO120 study. This study showed that the treatment more than doubled progression-free survival compared to palbociclib and fulvestrant alone, represents Roche’s initial targeted therapy for HR-positive disease, and incorporates a companion diagnostic to pinpoint eligible patients, thereby indicating a substantial advance in precision medicine for a patient population with limited first-line treatment options.
Who Are The Primary Competitors In The Global PD-L1 And PIK3CA Testing Market?
Major companies operating in the PD-L1 and PIK3CA testing market are F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories, Laboratory Corporation of America Holdings (Labcorp), Quest Diagnostics Incorporated, Eurofins Scientific SE, Agilent Technologies Inc., Sonic Healthcare Limited, Illumina Inc., Bio-Rad Laboratories Inc., QIAGEN N.V., Myriad Genetics Inc., NeoGenomics Laboratories Inc., BGI Genomics Co. Ltd., Guardant Health Inc., Caris Life Sciences, Foundation Medicine Inc., Amoy Diagnostics Co. Ltd., ACCB Biotech Private Limited, EntroGen Inc.
Download The Full Report For Exclusive Market Findings:
https://www.thebusinessresearchcompany.com/report/pd-l1-and-pik3ca-testing-global-market-report
How Is PD-L1 And PIK3CA Testing Market Demand Varying Across Different Regions?
North America was the largest region in the PD-L1 and PIK3CA testing market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in PD-L1 and PIK3CA testing report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East and Africa.
Request A Customized Version Of The PD-L1 And PIK3CA Testing Market Report:
https://www.thebusinessresearchcompany.com/customise?id=28289&type=smp
Browse Through More Reports Similar to the Global PD-L1 And PIK3CA Testing Market 2025, By The Business Research Company
Poc Hba1C Testing Market 2025
https://www.thebusinessresearchcompany.com/report/poc-hba1c-testing-market
Hpv Testing And Pap Test Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/hpv-testing-and-pap-test-global-market-report
D Dimer Testing Global Market Report 2025
https://www.thebusinessresearchcompany.com/report/d-dimer-testing-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model