Pediatric Clinical Trials Market Forecast 2026–2035: Data Trends and Industry Insights
Uncover key drivers, emerging technologies, and competitive movements shaping the pediatric clinical trials market from 2026–2035 with trusted insights from The Business Research Company
What level of growth is expected in the Pediatric Clinical Trials Market between 2026 and 2030?
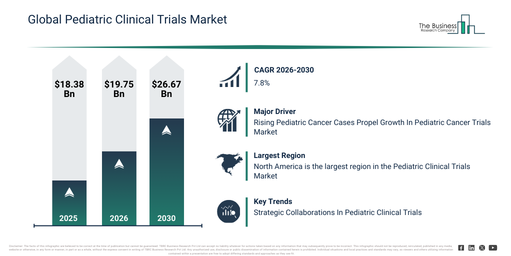
The pediatric clinical trials market size has experienced significant expansion in recent years. This market is projected to grow from $18.38 billion in 2025 to $19.75 billion in 2026, achieving a compound annual growth rate (CAGR) of 7.4%. The historical expansion observed within this market was influenced by elements such as constrained pediatric-specific drug research, a reliance on data from adult trials, inherent ethical and regulatory obstacles, difficulties in patient recruitment, and the limited utilization of advanced analytics in trial design.
The pediatric clinical trials market is set to experience robust expansion over the next few years. It is anticipated to grow to $26.67 billion by 2030, achieving a compound annual growth rate (CAGR) of 7.8%. This growth in the forecast period can be attributed to the rising demand for therapies tailored for children, the increased adoption of digital and AI-powered tools, growing regulatory incentives for pediatric studies, expanding collaborations between pharmaceutical companies and research institutions, and a heightened focus on patient-centric and personalized clinical trials. Major trends identified for this period include the increasing integration of precision medicine into pediatric clinical trials, the rising application of AI and digital technologies for trial design and data analysis, the growing incorporation of real-world evidence in pediatric research, increased investment in the development of pediatric drugs, and a heightened emphasis on personalized and age-specific therapeutic approaches.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=10350&type=smp
Which Drivers Are Expected To Impact The Pediatric Clinical Trials Market During The Forecast Period?
An anticipated increase in pediatric cancer cases is set to drive growth in the pediatric clinical trials market in the future. Pediatric cancer is defined as malignancies that emerge in children and young people below the age of 14. These cancers are very rare, and their patterns of development and spread can differ from adult tumors. Pediatric clinical trials remain crucial for improving prognoses and therapeutic approaches. For example, the American Cancer Society, a voluntary health organization in the US, reported in January 2023 an estimation that approximately 9,910 children in the US under the age of 15 would be diagnosed with cancer, and around 1,040 children under the age of 15 were projected to succumb to cancer in 2023. Consequently, expanding healthcare spending is propelling the pediatric clinical trials market.
How Is The Pediatric Clinical Trials Market Divided Into Segments?
The pediatric clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV
2) By Study Design: Treatment Studies, Observational Studies
3) By Therapeutic Areas: Infectious Diseases, Oncology, Autoimmune Or Inflammatory Diseases, Respiratory Disorders, Mental Health Disorders, Other Therapeutic Areas
Subsegments:
1) By Phase I: First-in-Pediatrics Trials, Dose Escalation Studies, Safety And Tolerability Assessments
2) By Phase II: Efficacy Studies, Dose Optimization Trials, Pharmacokinetic Studies
3) By Phase III: Large-Scale Efficacy Trials, Comparative Studies, Long-Term Safety Studies
4) By Phase IV: Post-Marketing Surveillance, Long-Term Safety Monitoring, Effectiveness Studies In Real-World Settings
Which Trends Are Expected To Influence The Pediatric Clinical Trials Market In The Upcoming Years?
Leading organizations engaged in pediatric clinical investigations are concentrating on collaboration and alliances to offer dependable services to consumers. A strategic partnership denotes a formalized relationship between two or more commercial entities, typically established through one or more business agreements or contracts. For example, in September 2023, Strados Labs, a US-based supplier of smart biosensors for tracking respiration, joined forces with Ann & Robert H. Lurie Children’s Hospital, a US-based pediatric acute care children’s hospital, to conduct a clinical study for the RESP Biosensor. The RESP Biosensor is a wearable instrument that utilizes acoustic sensors to monitor lung sounds for asthma signs like coughing and wheezing. The objective of this clinical trial is to evaluate the efficacy of the RESP Biosensor in precisely detecting and observing asthma exacerbations in children.
Which Leading Companies Dominate The Pediatric Clinical Trials Market Share?
Major companies operating in the pediatric clinical trials market are Medpac Inc., Pharmaceutical Product Development Inc., ICON plc, Syneos Health Inc., QPS Holdings LLC, Pfizer Inc., IQVIA Inc., Premier Research, Labcorp – Laboratory Corporation of America Holdings, The Emmes Company LLC, Synteract Inc., Charles River Laboratories International Inc., Covance Inc., Bristol-Myers Squibb Company, GlaxoSmithKline plc, BioNTech SE, Moderna Inc., Parexel International Corporation, PRA Health Sciences, Wuxi AppTec, Merck & Co. Inc., AstraZeneca plc, Sanofi S.A., Johnson & Johnson, Eli Lilly and Company, Takeda Pharmaceutical Company Limited, C.H. Boehringer Sohn AG & Co. KG, AbbVie Inc., Teva Pharmaceutical Industries Ltd., Oracle Health sciences
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/pediatric-clinical-trials-global-market-report
How Does The Pediatric Clinical Trials Market Perform Across Major Global Regions?
North America was the largest region in the pediatric clinical trials market in 2025. The regions covered in the pediatric clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Pediatric Clinical Trials Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=10350&type=smp
Browse Through More Reports Similar to the Global Pediatric Clinical Trials Market 2026, By The Business Research Company
Clinical Trial Supplies Market Report 2026
https://www.thebusinessresearchcompany.com/report/clinical-trial-supplies-global-market-report
Pediatric Supplements Market Report 2026
https://www.thebusinessresearchcompany.com/report/pediatric-supplements-global-market-report
Virtual Clinical Trials Market Report 2026
https://www.thebusinessresearchcompany.com/report/virtual-clinical-trials-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model