Pharmaceutical Impurity Synthesis and Isolation Services Market – Opportunities, Share, Growth and Competitive Analysis and Forecast 2029
Claim your 20% discount on Global Market Reports with code ONLINE20. Limited time only.
#What Is The Projected Valuation Of The Pharmaceutical Impurity Synthesis and Isolation Services Market In The Coming Years?#_x000D_
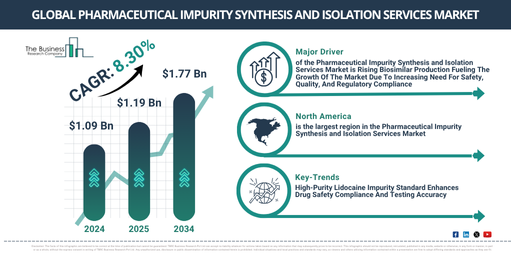
In the past few years, there has been a robust expansion in the market size of the pharmaceutical impurity synthesis and isolation services. The market, which stood at $1.09 billion in 2024, is projected to rise to $1.19 billion in 2025, registering a compound annual growth rate (CAGR) of 8.5%. The significant growth observed in the historic period is due to factors such as strict regulatory demands for impurity profiling, the heightened complexity of drug molecules, an uptick in the outsourcing from pharmaceutical firms, an increase in the production of generic drugs, and technological advancements in analytical instruments._x000D_
_x000D_
In the coming years, the market size for services related to pharmaceutical impurity synthesis and isolation is predicted to experience robust growth. The market value is projected to reach $1.63 billion in 2029, with a compound annual growth rate (CAGR) of 8.3%. This growth in the estimated period is credited to the expansion of biopharmaceutical pipelines, escalating demand for high-purity APIs, regulatory focus on nitrosamine and genotoxic impurities, progress in personalized medicine, and an upswing in investment in CRO/CDMO services. Some of the significant trends predicted for this period encompass the adoption of AI-fueled impurity identification, the incorporation of green chemistry in synthesis methods, an increase in global GMP-compliant service providers, the application of high-resolution mass spectrometry and NMR techniques, and strategic partnerships to expedite impurity profiling and isolation._x000D_
_x000D_
#Get A Free Sample Of The Report:#_x000D_
https://www.thebusinessresearchcompany.com/sample.aspx?id=25877&type=smp_x000D_
_x000D_
#What Drivers Are Accelerating Expansion Of The Pharmaceutical Impurity Synthesis and Isolation Services Market?#_x000D_
Accelerated production of biosimilars is predicted to stimulate the growth of the market for pharmaceutical impurity synthesis and isolation services. Biosimilars are biologic drugs which closely resemble an already approved reference product, and they show no noticeable differences in security, effectiveness, or quality. Due to the expiry of patents on original biologic drugs, the creation of biosimilars has increased, allowing producers to establish more cost-effective substitutes and widen treatment access. These services for the synthesis and isolation of pharmaceutical impurities play an essential role in biosimilar manufacturing by assisting in the identification, characterization, and management of impurities, thereby ensuring the biosimilar’s safety, quality, and adherence to regulatory standards. For example, as per the African Development Bank’s report in March 2025, XpandC, a private banking firm based in Cote d’Ivoire, is expected to amplify its annual biosimilar production capacity from 3 million to 7.65 million doses by 2032 and will add two more biosimilars. Consequently, the escalating production of biosimilars is propelling the growth of the market for pharmaceutical impurity synthesis and isolation services._x000D_
_x000D_
#How Is The Global Pharmaceutical Impurity Synthesis and Isolation Services Market Broken Down By Segment?#_x000D_
The pharmaceutical impurity synthesis and isolation services market covered in this report is segmented – _x000D_
_x000D_
1) By Service: Synthesis Services, Isolation Services, Analytical Services_x000D_
2) By Impurity Type: Organic Impurities, Inorganic Impurities, Residual Solvents_x000D_
3) By Technique: Chromatography, Spectroscopy, Crystallization, Hyphenated Techniques, Other Techniques_x000D_
4) By Application: Drug Development, Commercial Manufacturing, Quality Control, Regulatory Compliance_x000D_
5) By End User: Biotech And Pharmaceutical Companies, Contract Research Organizations (CRO), Other End Users_x000D_
_x000D_
Subsegments:_x000D_
1) By Synthesis Services: Custom Impurity Synthesis, Stable Isotope-Labeled Impurity Synthesis, Process-Related Impurity Synthesis, Degradation Product Synthesis, Metabolite Synthesis_x000D_
2) By Isolation Services: Isolation Of Process Impurities, Isolation Of Degradation Impurities, Preparative Chromatography-Based Isolation, Crystallization-Based Isolation, Flash Chromatography Isolation_x000D_
3) By Analytical Services: Impurity Profiling, Structural Elucidation, Quantitative Analysis, Genotoxic Impurity Analysis, Stability Studies_x000D_
_x000D_
#Which Trends Are Likely To Redefine Growth Paths In The Pharmaceutical Impurity Synthesis and Isolation Services Market?#_x000D_
Leading firms in the pharmaceutical impurity synthesis and isolation services market are directing their efforts towards devising unique solutions like lidocaine impurity scrutiny and validation. The plan is to ascertain drug safety, adhere to regulatory compliance, and augment the quality of their pharmaceutical products. The procedure of lidocaine impurity validation and testing aims at detection and quantification of impurities present in lidocaine products to affirm their quality, safety, and consistency with regulatory guidelines. For example, Advent Pharma Limited, a company rooted in Bangladesh, unveiled a high-purity reference benchmark for a significant lidocaine impurity called 1,4-Bis(2,6-dimethylphenyl) piperazine-2,5-dione, in July 2025. This impurity, a result of either lidocaine synthesis or degradation, demands regular check-ups to assure the drug’s safety and effectiveness. The standard delivered by Advent, with its purity hitting =97%, is served with comprehensive analytical validation and tailored to fulfil strict regulatory conditions like ICH Q3A/B and Q2 instructions._x000D_
_x000D_
#Which Firms Are Making The Biggest Impact In The Pharmaceutical Impurity Synthesis and Isolation Services Market?#_x000D_
Major companies operating in the pharmaceutical impurity synthesis and isolation services market are Thermo Fisher Scientific Inc., Merck KGaA, Laboratory Corporation of America Holdings, SGS SA, Eurofins Scientific SE, Agilent Technologies Inc., WuXi AppTec Co. Ltd., Catalent Inc., Charles River Laboratories International Inc., Intertek Group plc, Waters Corporation, Almac Group Limited, Piramal Pharma Limited, Cambrex Corporation, Syngene International Limited, Frontage Laboratories Inc., Pharmaron Beijing Co. Ltd., Symeres B.V., Synergenics Canada Inc., Veeda Clinical Research Limited, Alfa Chemistry LLC, Epichem Pty Ltd., Creative Dynamics Inc., PCI Pharma Services LLC, and VEEPRHO s.r.o. _x000D_
_x000D_
#Access The Complete Report Here:#_x000D_
_x000D_
#Which Geographic Regions Are Creating Strong Demand In The Pharmaceutical Impurity Synthesis and Isolation Services Market?#_x000D_
North America was the largest region in the pharmaceutical impurity synthesis and isolation services market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmaceutical impurity synthesis and isolation services market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa._x000D_
_x000D_
#Customize Your Report Here:#_x000D_
https://www.thebusinessresearchcompany.com/customise?id=25877&type=smp_x000D_
_x000D_
Browse Through More Reports Similar to the Global Pharmaceutical Impurity Synthesis and Isolation Services Market 2025, By The Business Research Company_x000D_
_x000D_
Pharmaceutical Intermediates Global Market Report 2025_x000D_
_x000D_
Pharmaceutical Processing Seals Global Market Report 2025_x000D_
_x000D_
Advanced Therapeutics Pharmaceutical Outsourcing Global Market Report 2025_x000D_
_x000D_
#About The Business Research Company:#_x000D_
_x000D_
With over 17500+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game._x000D_
_x000D_
#Get in touch with us:#_x000D_
_x000D_
The Business Research Company: https://www.thebusinessresearchcompany.com/_x000D_
Americas +1 310-496-7795_x000D_
Asia +44 7882 955267 & +91 8897263534_x000D_
Europe +44 7882 955267_x000D_
Email us at info@tbrc.info_x000D_
_x000D_
#Follow us on:#_x000D_
_x000D_
LinkedIn: https://in.linkedin.com/company/the-business-research-company_x000D_
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ_x000D_
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model