Pharmaceutical Impurity Synthesis and Isolation Services Market Forecast 2026–2030 Presenting Long-Term Industry Insights
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
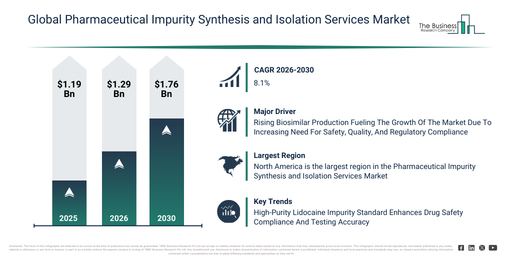
By 2030, What Market Size Is The Pharmaceutical Impurity Synthesis and Isolation Services Market Expected To Reach Based On Its 2026 Value?
The pharmaceutical impurity synthesis and isolation services market size has witnessed significant expansion in recent years. This market is projected to escalate from $1.19 billion in 2025 to $1.29 billion in 2026, exhibiting a compound annual growth rate (CAGR) of 8.4%. The historical increase in this market can be attributed to several factors including the limited availability of specialized impurity synthesis services, a reliance on conventional analytical techniques, increasing pharmaceutical manufacturing activities, growing regulatory scrutiny, and the adoption of basic chromatographic methods.
The pharmaceutical impurity synthesis and isolation services market is poised for significant expansion in the coming years. It is projected to reach $1.76 billion by 2030, demonstrating an impressive compound annual growth rate (CAGR) of 8.1%. This anticipated growth is primarily driven by advancements in hyphenated analytical techniques, the broadening scope of metabolite and degradation product synthesis, increasing outsourcing to cros, the rising adoption of stability studies, and the growth in regulatory-driven impurity profiling. Furthermore, key trends for this period include a rising demand for custom impurity synthesis services, the increasing adoption of advanced isolation techniques, an expansion in analytical services for regulatory compliance, the growth of drug development and quality control programs, and the integration of stability and genotoxic impurity analysis.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25877&type=smp
Which Major Drivers Are Influencing The Expansion Of The Pharmaceutical Impurity Synthesis and Isolation Services Market?
The increasing output of biosimilars is projected to stimulate the expansion of the pharmaceutical impurity synthesis and isolation services market in the future. Biosimilars are defined as biologic medications closely resembling an already authorized reference product, exhibiting no notable variances in their safety, efficacy, or overall quality. The climb in biosimilar production stems from the expiration of patents on original biologic drugs, which allows producers to develop more cost-effective options and broaden treatment accessibility. Pharmaceutical impurity synthesis and isolation services prove beneficial for biosimilar manufacturing through their role in identifying, characterizing, and managing impurities, thereby guaranteeing the biosimilar’s safety, quality, and adherence to regulations. As an illustration, in March 2025, the African Development Bank, a private banking entity based in Cote d’Ivoire, reported that the new XpandC facility is poised to elevate annual biosimilar production capacity from 3 million to 7.65 million doses by 2032, alongside the introduction of two additional biosimilars. Consequently, the escalating production of biosimilars is a key factor propelling the expansion of the pharmaceutical impurity synthesis and isolation services market.
What Segment Categories Shape The Pharmaceutical Impurity Synthesis and Isolation Services Market Segment Landscape?
The pharmaceutical impurity synthesis and isolation services market covered in this report is segmented –
1) By Service: Synthesis Services, Isolation Services, Analytical Services
2) By Impurity Type: Organic Impurities, Inorganic Impurities, Residual Solvents
3) By Technique: Chromatography, Spectroscopy, Crystallization, Hyphenated Techniques, Other Techniques
4) By Application: Drug Development, Commercial Manufacturing, Quality Control, Regulatory Compliance
5) By End User: Biotech And Pharmaceutical Companies, Contract Research Organizations (CRO), Other End Users
Subsegments:
1) By Synthesis Services: Custom Impurity Synthesis, Stable Isotope-Labeled Impurity Synthesis, Process-Related Impurity Synthesis, Degradation Product Synthesis, Metabolite Synthesis
2) By Isolation Services: Isolation Of Process Impurities, Isolation Of Degradation Impurities, Preparative Chromatography-Based Isolation, Crystallization-Based Isolation, Flash Chromatography Isolation
3) By Analytical Services: Impurity Profiling, Structural Elucidation, Quantitative Analysis, Genotoxic Impurity Analysis, Stability Studies
Which Trends Are Guiding The Direction Of The Pharmaceutical Impurity Synthesis and Isolation Services Market?
Leading companies in the pharmaceutical impurity synthesis and isolation services market are prioritizing the creation of advanced solutions, such as lidocaine impurity validation and testing, to secure drug safety, enhance adherence to regulations, and elevate the overall quality of pharmaceutical products. Lidocaine impurity validation and testing refers to the systematic process of identifying and quantifying impurities within lidocaine products to confirm their safety, quality, and compliance with governing standards. For example, in July 2025, Advent Pharma Limited, a Bangladesh-based pharmaceutical company, unveiled a high-purity reference standard for a critical lidocaine impurity, 1,4-Bis(2,6-dimethylphenyl) piperazine-2,5-dione, intended to aid pharmaceutical firms in method validation, stability testing, and regulatory filings for lidocaine formulations. This impurity, which can emerge during the manufacturing or degradation of lidocaine, demands routine scrutiny to maintain drug safety and efficacy. Advent’s standard, possessing a purity of =97%, comes with extensive analytical documentation and is engineered to fulfill strict regulatory stipulations, including ICH Q3A/B and Q2 guidelines.
Who Are The Companies Operating Across The Pharmaceutical Impurity Synthesis and Isolation Services Market Value Chain?
Major companies operating in the pharmaceutical impurity synthesis and isolation services market are Thermo Fisher Scientific Inc., Merck KGaA, Laboratory Corporation of America Holdings, SGS SA, Eurofins Scientific SE, Agilent Technologies Inc., WuXi AppTec Co. Ltd., Catalent Inc., Charles River Laboratories International Inc., Intertek Group plc, Waters Corporation, Almac Group Limited, Piramal Pharma Limited, Cambrex Corporation, Syngene International Limited, Frontage Laboratories Inc., Pharmaron Beijing Co. Ltd., Symeres B.V., Synergenics Canada Inc., Veeda Clinical Research Limited, Alfa Chemistry LLC, Epichem Pty Ltd., PCI Pharma Services LLC, and VEEPRHO s.r.o.
Get The Full Pharmaceutical Impurity Synthesis and Isolation Services Market Report:
Where Is The Pharmaceutical Impurity Synthesis and Isolation Services Market Most Concentrated Geographically?
North America was the largest region in the pharmaceutical impurity synthesis and isolation services market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the pharmaceutical impurity synthesis and isolation services market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Pharmaceutical Impurity Synthesis and Isolation Services Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Pharmaceutical Impurity Synthesis and Isolation Services Market 2026, By The Business Research Company
Pharmaceutical Isolator Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharmaceutical-isolator-global-market-report
Pharmaceutical Filtration Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharmaceutical-filtration-global-market-report
Sterile Compounding Pharmacies Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at: marketing@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model