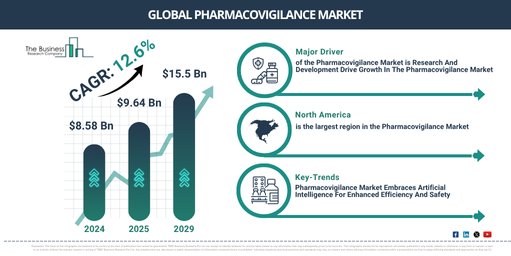
How Will The Pharmacovigilance Market Expand At A CAGR Of 12.6% Through 2029?
Claim 20% Off Global Market Reports With Code ONLINE20 – Insights on Tariff Effects, Global Trade, and Industry Disruptors
How Is The Pharmacovigilance Market Expected To Grow In Terms Of Size?
Over the past few years, the pharmacovigilance market size has witnessed substantial growth. It is predicted that this market will expand from $8.58 billion in 2024 to reach $9.64 billion in 2025, exhibiting a compound annual growth rate (CAGR) of 12.4%. Factors contributing to historical growth include the rising cases of adverse drug reactions (ADRs), the surge in research and development (R&D) activities, the increasing rate of drug development, a heightened emphasis on personalized medicine, and an escalating prevalence of chronic diseases.
The market size of pharmacovigilance is set to witness substantial expansion in the coming years and is projected to reach “$15.5 billion by 2029, with a compound annual growth rate (CAGR) of 12.6%. The significant growth anticipated during this period is due to factors such as increased healthcare spending, enhanced government assistance, and an aging population. Prevailing trends for the forecast period encompass escalated investments, utilization of artificial intelligence, adoption of inventive platforms, implementation of cutting-edge cloud-based systems, and strategic collaborations and acquisitions.
Get A Free Sample Of The Report:
https://www.thebusinessresearchcompany.com/sample.aspx?id=5928&type=smp
What Market Forces Are Contributing To The Growth Of The Pharmacovigilance Market?
The growth of the pharmacovigilance market is fuelled by escalating investment in research and development activities. The enlargement of new and inventive drugs with enhanced safety levels is made possible by this R&D expenditure. Comprehensive examinations during the developmental phase of the drug enable the early recognition and management of prospective safety risks. Furthermore, progress in technology for adverse event tracing, data scrutinizing, and signal detection catalysed by R&D initiatives facilitates the timely discovery of adverse drug responses in the post-marketing phase. For example, as reported by the Llywodraeth Cymru Welsh Government, a government agency in the UK, in February 2024, £1.0 billion ($1.25 billion), representing 1.9% of the total UK’s research and development expenditure was invested by Wales on Business Expenditure on Research and Development (BERD) in 2022. Consequently, the heightened research activities significantly contributed to the expansion of the pharmacovigilance market.
What Are The Primary Segmentation Parameters In The Pharmacovigilance Market?
The pharmacovigilance market covered in this report is segmented –
1) By Type: Spontaneous Reporting, Intensified ADR Reporting, Targeted Spontaneous Reporting, Cohort Event Monitoring, EHR Mining
2) By Service Provider: In-House, Contract Outsourcing
3) By Process Flow: Case Data Management, Signal Detection, Risk Management System
4) By Clinical Trial Phases: Preclinical, Phase I, Phase II, Phase III, Phase IV
5) By End User: Hospitals, Pharmaceutical Companies, Other End Users
Subsegments:
1) By Spontaneous Reporting: Individual Case Safety Reports (ICSRs), Voluntary Reporting Systems, Case Reports From Healthcare Professionals
2) By Intensified ADR Reporting: Active Surveillance Programs, Enhanced Monitoring In Clinical Trials, Risk Minimization Strategies
3) By Targeted Spontaneous Reporting: Reporting For Specific Drug Classes, Condition-Specific Reporting, Focused Surveillance Programs
4) By Cohort Event Monitoring: Prospective Cohort Studies, Retrospective Cohort Studies, Longitudinal Studies
5) By EHR Mining: Data Extraction From Electronic Health Records, Signal Detection Through EHR Analysis, Integration Of EHR Data With Pharmacovigilance Systems
Which Emerging Trends Are Reshaping The Pharmacovigilance Market Landscape?
Pharmacovigilance market players are turning their attention towards artificial intelligence (AI) to bolster efficiency. AI has the potential to enhance both the effectiveness and precision of pharmacovigilance tasks, which could lead to superior patient results. Use of AI allows pharmaceutical companies to streamline operations, enhance data handling, speed up drug discovery, while concurrently lowering expenses and amplifying safety. For example, in August 2024, the Indian Government initiated the Adverse Drug Reaction Monitoring System (ADRMS) software, launched by the Minister of Health & Family Welfare and the Minister of Chemicals and Fertilizers. This bespoke pharmacovigilance software simplifies the gathering and evaluation of adverse incidents relating to drugs and medical apparatus, making the reporting protocol less complex for both patients and healthcare professionals. This drive notably bolsters India’s pharmacovigilance foundations by ensuring a more complete acquisition of safety findings and bolstering medication safety observation. The ADRMS is geared towards enhancing the effectiveness of drug safety management and compliance to norms, aiding India’s progression as a frontrunner in the international pharmaceutical sector.
Which Players Are Shaping The Competitive Landscape Of The Pharmacovigilance Market?
Major companies operating in the pharmacovigilance market include IQVIA, Cognizant, ICON Plc, Accenture plc, PAREXEL International Corporation., United BioSource LLC, ArisGlobal, Quanticate, Wipro Limited, Linical Americas, Novotech CRO, Wuxi Apptec, Simcere Pharmaceutical Group, Lee’s Pharmaceutical Holdings, Luye Pharma Group, EXTEDO, Arriello, PrimeVigilance, Axios International, C3i Solutions, Alcon, Secure AI Labs, OmniSol dot Tech, Canna Call Company, Veripad, Prevnos Inc., Lifescient, Inc, BeiGene, QuintilesIMS, Labcorp Drug Development, Pharmaceutical Product Development Inc., PRA Health Sciences, Synowledge LLC, RAPAhub, illuminate health, Invenio Medical, ClinChoice Inc., MSD, Novasyte, Fortrea Holdings Inc, Syneos Health Clinical, Zoetis Inc., Telerx Marketing Inc., Gilead Sciences, Inc., Elanco, Abbott Laboratories, Spimaco, Teva Pharmaceutical Industries Ltd.
Access The Complete Report Here:
https://www.thebusinessresearchcompany.com/report/pharmacovigilance-global-market-report
What Are The Emerging Regional Trends Driving The Pharmacovigilance Market?
North America was the largest region in the pharmacovigilance market in 2024. Asia-Pacific is expected to be the fastest-growing region in the forecast region. The regions covered in the pharmacovigilance market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Customize Your Report Here:
https://www.thebusinessresearchcompany.com/customise?id=5928&type=smp
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model