Pharmacovigilance Market Forecast 2026–2035: Data Trends and Industry Insights
Uncover key drivers, emerging technologies, and competitive movements shaping the pharmacovigilance market from 2026–2035 with trusted insights from The Business Research Company
What upper market value could the Pharmacovigilance Market reach by 2030 starting from 2026 levels?
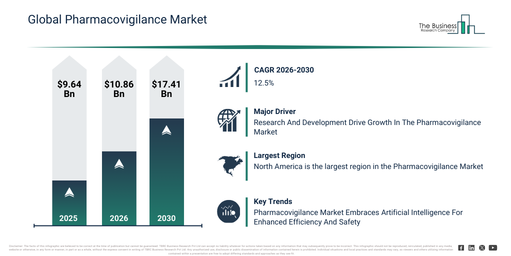
The pharmacovigilance market size has seen rapid expansion in recent years. It is anticipated to increase from $9.64 billion in 2025 to $10.86 billion in 2026, achieving a compound annual growth rate (CAGR) of 12.7%. The historical growth of this market can be attributed to several factors, including a rise in adverse drug reaction reporting, the broadening of global pharmaceutical approvals, an increase in clinical trial volumes, the establishment of international drug safety regulations, and the growing usage of drugs for chronic diseases.
The pharmacovigilance market is projected to experience substantial expansion over the coming years, reaching $17.41 billion by 2030, driven by a compound annual growth rate (CAGR) of 12.5%. This anticipated growth during the forecast period is fueled by several factors, including the increased use of real-world data, a rise in approvals for biologics and biosimilars, the growing intricacy of combination therapies, the expansion of pharmacovigilance activities in developing economies, and a heightened emphasis on patient safety results. Key trends anticipated during this period encompass the broadening of post-marketing surveillance efforts, the increasing incorporation of real-world evidence into safety monitoring, intensified regulatory oversight and adherence demands, a surge in the outsourcing of pharmacovigilance services, and the merging of multisource safety data platforms.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=5928&type=smp
Which Drivers Are Influencing Long-Term Growth In The Pharmacovigilance Market?
The expansion of research and development endeavors is poised to stimulate the pharmacovigilance market. By allocating resources to R&D, pharmaceutical enterprises are able to formulate new and innovative medicines with superior safety profiles. Rigorous testing carried out during the preclinical and clinical stages of drug development facilitates the early identification of potential safety concerns, allowing for the implementation of appropriate risk mitigation plans. Moreover, R&D efforts contribute to the advancement of techniques for monitoring adverse events, analyzing data, and detecting signals, which in turn supports the prompt discovery of adverse drug reactions in the post-marketing phase. For instance, in 2023, according to Office for National Statistics, a UK-based, government agency, the UK government’s net spending on research and development (R&D) increased to £17.4 billion in 2023, up from £16.1 billion in 2022, marking an 8.2% rise. Thus, the escalating research and development activities have underpinned the expansion of the pharmacovigilance market.
Which Segments Are Driving Activity In The Pharmacovigilance Market?
The pharmacovigilance market covered in this report is segmented –
1) By Type: Spontaneous Reporting, Intensified ADR Reporting, Targeted Spontaneous Reporting, Cohort Event Monitoring, EHR Mining
2) By Service Provider: In-House, Contract Outsourcing
3) By Process Flow: Case Data Management, Signal Detection, Risk Management System
4) By Clinical Trial Phases: Preclinical, Phase I, Phase II, Phase III, Phase IV
5) By End User: Hospitals, Pharmaceutical Companies, Other End Users
Subsegments:
1) By Spontaneous Reporting: Individual Case Safety Reports (ICSRs), Voluntary Reporting Systems, Case Reports From Healthcare Professionals
2) By Intensified ADR Reporting: Active Surveillance Programs, Enhanced Monitoring In Clinical Trials, Risk Minimization Strategies
3) By Targeted Spontaneous Reporting: Reporting For Specific Drug Classes, Condition-Specific Reporting, Focused Surveillance Programs
4) By Cohort Event Monitoring: Prospective Cohort Studies, Retrospective Cohort Studies, Longitudinal Studies
5) By EHR Mining: Data Extraction From Electronic Health Records, Signal Detection Through EHR Analysis, Integration Of EHR Data With Pharmacovigilance Systems
What Long-Term Trends Are Expected To Shape The Future Of The Pharmacovigilance Market?
Leading enterprises within the pharmacovigilance market are increasingly leveraging artificial intelligence (AI) to enhance their operational effectiveness. AI has the capacity to boost both the efficiency and precision of pharmacovigilance operations, ultimately contributing to superior patient results. Through the deployment of AI, pharmaceutical firms can optimize workflows, refine data handling, and expedite drug discovery, all while simultaneously cutting expenses and elevating safety standards. As an illustration, in August 2024, the Government of India rolled out the Adverse Drug Reaction Monitoring System (ADRMS) software, which was unveiled by the Minister of Health & Family Welfare and Minister of Chemicals and Fertilizers. This dedicated pharmacovigilance software aids in the gathering and examination of adverse events linked to pharmaceuticals and medical apparatus, thereby simplifying the reporting mechanism for both end-users and medical practitioners. This undertaking substantially bolsters India’s pharmacovigilance framework by guaranteeing a more exhaustive collection of safety data and augmenting drug safety surveillance. The ADRMS seeks to elevate the effectiveness of drug safety supervision and adherence to regulations, thereby assisting India in establishing itself as a frontrunner in the worldwide pharmaceutical sector.
Which Leading Companies Dominate The Pharmacovigilance Market Share?
Major companies operating in the pharmacovigilance market are IQVIA, Cognizant, ICON Plc, Accenture plc, PAREXEL International Corporation, United BioSource LLC, ArisGlobal, Quanticate, Wipro Limited, Linical Americas, Novotech CRO, EXTEDO, Arriello, PrimeVigilance, Axios International, C3i Solutions, Secure AI Labs, OmniSol dot Tech, Prevnos Inc., Lifescient Inc, QuintilesIMS, Labcorp Drug Development, Pharmaceutical Product Development Inc., PRA Health Sciences, Synowledge LLC, RAPAhub, illuminate health, Invenio Medical, ClinChoice Inc., Novasyte, Fortrea Holdings Inc., Syneos Health Clinical, Telerx Marketing Inc.
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/pharmacovigilance-global-market-report
How Does The Pharmacovigilance Market Perform Across Major Global Regions?
North America was the largest region in the pharmacovigilance market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast region. The regions covered in the pharmacovigilance market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Pharmacovigilance Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=5928&type=smp
Browse Through More Reports Similar to the Global Pharmacovigilance Market 2026, By The Business Research Company
Pharmacovigilance Market Report 2026
https://www.thebusinessresearchcompany.com/report/pharmacovigilance-global-market-report
Medical Devices Vigilance Market Report 2026
https://www.thebusinessresearchcompany.com/report/medical-devices-vigilance-global-market-report
Cosmetovigilance Market Report 2026
https://www.thebusinessresearchcompany.com/report/cosmetovigilance-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model