Point-Of-Care Molecular Diagnostics Market Future Outlook Showing Expansion Toward $7.91 Billion by 2030
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Market Size Increase Is Anticipated For The Point-Of-Care Molecular Diagnostics Market Over The 2026–2030 Period?
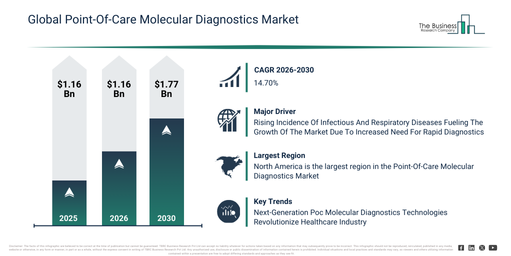
The point-of-care molecular diagnostics market size has seen rapid growth in recent years. This market is expected to expand from $3.98 billion in 2025 to $4.58 billion in 2026, at a compound annual growth rate (CAGR) of 15.0%. Historically, this expansion has been driven by factors including the increasing prevalence of infectious diseases, the wider availability of emergency diagnostic testing, a growing demand for rapid clinical decision support, continuous improvements in molecular assay technologies, and the increasing utilization of point-of-care diagnostics in hospitals.
The point-of-care molecular diagnostics market is projected for swift expansion in the coming years. Its valuation is anticipated to reach $7.91 billion by 2030, driven by a compound annual growth rate (CAGR) of 14.7%. This expansion during the projection period stems from factors such as the greater acceptance of home molecular testing, increased funding in rapid diagnostic facilities, broader uses for respiratory and oncology tests, expanding integration of cloud-based diagnostic systems, and a sharpened focus on detecting diseases earlier. Key developments anticipated within the forecast timeframe encompass the wider embrace of rapid molecular testing platforms, a heightened need for decentralized diagnostic approaches, increasing incorporation of automated sample handling, the broadening of syndromic testing panels, and an improved emphasis on quick result delivery.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=7046&type=smp
Which Major Factors Are Driving The Expansion Of The Point-Of-Care Molecular Diagnostics Market?
The swift increase in the occurrence of infectious and respiratory diseases is anticipated to notably fuel the expansion of the point-of-care molecular diagnostics market. Respiratory infections, such as viruses or bacteria, stem from microorganisms that affect the respiratory system. These microbes can spread via coughing, sneezing, or direct physical contact. The management of severe respiratory conditions involves various inhalers, oral medications, intravenous treatments, and point-of-care molecular diagnostic methods to treat patients. For instance, in June 2024, as reported by the UK Health Security Agency, a UK-based government agency, between week 40 of 2022 and week 15 of 2023, 108 NHS trusts in England documented 1,681 critical care admissions for confirmed influenza. In contrast, the preceding season recorded 182 admissions across 114 trusts, escalating to 316 by week 39 of 2022. Hence, the rapid incidence of infectious and respiratory diseases and associated mortality will propel the point-of-care molecular diagnostics market.
How Is The Point-Of-Care Molecular Diagnostics Market Segmented Across Key Categories?
The point-of-care molecular diagnostics market covered in this report is segmented –
1) By Product: Assays And Kits, Instruments And Analyzers
2) By Application: Respiratory Infections, Gastrointestinal Infections, Sexually Transmitted Infections (STIs), Hospital-Acquired Infections (HAIs), Bloodstream and Sepsis-Related Infections
3) By End-User: Decentralized Labs, Hospitals, Home Care, Assisted Living Healthcare Facilities
Subsegments:
1) By Assays And Kits: Nucleic Acid Amplification Tests (NAATs), Rapid Diagnostic Tests, Molecular Detection Kits, Reagents And Consumables
2) By Instruments And Analyzers: Microfluidic Devices, Point-of-Care Analyzers
Which Upcoming Trends Are Expected To Influence The Point-Of-Care Molecular Diagnostics Market?
Leading companies in the point-of-care molecular diagnostics market are concentrating on providing technologically advanced solutions to bolster their market position. These organizations are deploying next-generation point-of-care molecular diagnostics technologies and associated services, such as enzyme-linked immunosorbent assay (ELISA), polymerase chain reaction (PCR), mass spectrometry (MS), situ hybridizations, spectral karyotyping imaging, and DNA microarrays, among others. These advancements are being implemented on POC devices to accelerate analysis times and lower costs. For instance, in May 2023, Sensible Diagnostics, a US-based diagnostics company, introduced a 10-minute point-of-care PCR system. Sensible Diagnostics is leveraging the expertise Curative gained from its diagnostics business during the pandemic. The firm stated it has performed more than two million molecular point-of-care COVID-19 tests, and its PCR platform boasts a 10-minute time-to-result.
Who Are The Dominant Players Shaping The Point-Of-Care Molecular Diagnostics Market Landscape?
Major companies operating in the point-of-care molecular diagnostics market are Abbott Laboratories, F Hoffmann La Roche Ltd, BioMérieux SA, Hologic Inc, Becton Dickinson and Company, Siemens Healthineers AG, QuidelOrtho Corporation, Bio Rad Laboratories Inc, Qiagen N V, Biocartis Group NV, DiaSorin S p A, SD Biosensor Inc, Cue Health Inc, Visby Medical Inc, Binx Health Inc, Molbio Diagnostics Pvt Ltd, Credo Diagnostics Biomedical Pvt Ltd, Genetron Health, Mammoth Biosciences Inc, Seegene Inc
Read the full point-of-care molecular diagnostics market report here:
Which Region Currently Holds The Largest Share Of The Point-Of-Care Molecular Diagnostics Market?
North America was the largest region in the point-of-care molecular diagnostics market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the point-of-care molecular diagnostics market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Point-Of-Care Molecular Diagnostics Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=7046&type=smp
Browse Through More Reports Similar to the Global Point-Of-Care Molecular Diagnostics Market 2026, By The Business Research Company
Molecular Diagnostics Devices And Equipment Market Report 2026
Point Of Care Diagnostics Market Report 2026
https://www.thebusinessresearchcompany.com/report/point-of-care-diagnostics-global-market-report
Biomarker Clinical Phase Outsourcing Services Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model