Post-Marketing Pharmacovigilance And Medical Information Market Forecast With Insights On Demand And Industry Growth
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
How Much Will The Market Value Of The Post-Marketing Pharmacovigilance And Medical Information Market Grow Between 2026 And 2030?
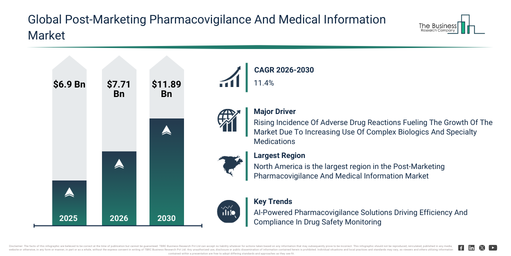
The market for post-marketing pharmacovigilance and medical information has experienced rapid expansion recently. It is projected to increase from $6.9 billion in 2025 to $7.71 billion by 2026, demonstrating a compound annual growth rate (CAGR) of 11.7%. Historically, this growth can be linked to factors such as inadequate post-marketing surveillance systems, dependence on manual adverse drug reaction (ADR) submissions, evolving regulatory demands for drug safety, a rise in new drug authorizations, and the absence of unified hospital data platforms.
The post-marketing pharmacovigilance and medical information market is projected for significant expansion in the coming years. Its value is anticipated to reach $11.89 billion by 2030, advancing at a compound annual growth rate (CAGR) of 11.4%. This anticipated growth is driven by factors such as the incorporation of AI and ML algorithms for signal detection, the utilization of longitudinal patient data analysis, the broader scope of disease registry-linked surveillance, a surge in digital application-based reporting, and increased funding for real-time pharmacovigilance solutions. Key developments foreseen during this period encompass the growing uptake of electronic health record (EHR) data mining, the wider implementation of real-time adverse drug reaction (ADR) monitoring systems, an increase in reporting tailored to specific risk populations and therapeutic classes, the extension of cohort event monitoring for novel drug users, and the unification of digital and mobile platforms for pharmacovigilance reporting.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=27393&type=smp
What Key Factors Are Shaping The Post-Marketing Pharmacovigilance And Medical Information Market Landscape?
The increasing occurrence of adverse drug reactions is anticipated to fuel the expansion of the post-marketing pharmacovigilance and medical information market moving forward. Adverse drug reactions are defined as undesirable or damaging effects that manifest when a medicine is administered at its recommended dosage for the purpose of prevention, diagnosis, or treatment. The frequency of adverse drug reactions is escalating as the aging demographic increasingly requires treatment for numerous chronic conditions, leading to the simultaneous use of several medications, which heightens the probability of harmful drug interactions and unexpected side effects. Post-marketing pharmacovigilance and medical information aid in managing adverse drug reactions by identifying, tracking, and sharing information on potential side effects after a medicine has been launched, thereby helping to maintain its safety and effectiveness. For instance, in January 2023, according to Navikenz, a US-based artificial intelligence-focused IT services company, in the US, adverse drug reactions are estimated to cause 200,000 to 400,000 deaths annually, surpassing the combined fatalities from stroke and diabetes, with approximately 2.7 million cases each year resulting in over 100,000 hospitalizations and more than 15,000 deaths. Thus, the escalating incidence of adverse drug reactions is driving the growth of the post-marketing pharmacovigilance and medical information market.
How Is The Post-Marketing Pharmacovigilance And Medical Information Market Broken Down By Segment Categories?
The post-marketing pharmacovigilance and medical information market covered in this report is segmented –
1) By Type: Spontaneous Reporting, Intensified Adverse Drug Reaction (ADR) Reporting, Targeted Spontaneous Reporting, Cohort Event Monitoring, Electronic Health Record (EHR) Mining
2) By Publication Type: Books, Online Media, Journals
3) By End User: Hospitals, Research Organizations, Other End-Users
Subsegments:
1) By Spontaneous Reporting: Consumer Reporting, Healthcare Professional Reporting, Regulatory Authority Reporting, Pharmaceutical Company Reporting, Digital App-Based Reporting
2) By Intensified Adverse Drug Reaction (ADR) Reporting: Hospital-Based Surveillance, Program-Specific Monitoring, Disease-Specific Monitoring, Product-Specific Monitoring, Real-Time Monitoring Systems
3) By Targeted Spontaneous Reporting: Risk Population-Based Reporting, Therapeutic Class-Based Reporting, Adverse Drug Reaction Focused Reporting, Region-Specific Reporting, Healthcare Setting-Specific Reporting
4) By Cohort Event Monitoring: Prospective Cohort Monitoring, Retrospective Cohort Monitoring, Active Follow-Up Monitoring, New Drug User Monitoring, Disease Registry-Linked Monitoring
5) By Electronic Health Record (EHR) Mining: Natural Language Processing-Based Mining, Artificial Intelligence (AI) And Machine Learning Algorithms, Rule-Based Signal Detection, Longitudinal Patient Data Analysis, Integrated Hospital Data Systems
What Trends Are Influencing The Post-Marketing Pharmacovigilance And Medical Information Market?
Leading companies within the post-marketing pharmacovigilance and medical information market are prioritizing the development of advanced solutions, such as AI-powered pharmacovigilance workflows, to improve the accuracy of adverse event detection and enable real-time safety monitoring, ultimately leading to better patient outcomes. These AI-powered pharmacovigilance workflows utilize artificial intelligence to automate drug safety surveillance, facilitating quicker adverse event identification and enhancing regulatory compliance. For instance, in January 2024, PubHive Ltd., a UK-based cloud-based software-as-a-service (SaaS) company, introduced a centralized Summary of Product Characteristics (SmPC) management system for life science companies and research organizations. This platform consolidates essential drug safety information into a single, readily available repository, automates literature monitoring and regulatory reporting, strengthens team collaboration, and streamlines compliance processes through sophisticated AI capabilities. This empowers pharmacovigilance teams to operate with increased efficiency and proactivity, ensuring both patient safety and adherence to regulatory standards.
Which Players Are Present In The Post-Marketing Pharmacovigilance And Medical Information Market Space?
Major companies operating in the post-marketing pharmacovigilance and medical information market are Accenture plc, Sanofi S.A., Capgemini SE, Merck & Co. Inc., Cognizant Technology Solutions Corporation, IQVIA Holdings Inc., HCL Technologies Limited, ICON plc, Syneos Health Inc., Genpact Limited, Parexel International Corporation, Avalere Health LLC, Quanticate Limited, Inizio Consulting LLC
Get The Full Post-Marketing Pharmacovigilance And Medical Information Market Report:
Which Region Holds The Highest Market Share In The Post-Marketing Pharmacovigilance And Medical Information Market?
North America was the largest region in the post-marketing pharmacovigilance and medical information market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the post-marketing pharmacovigilance and medical information market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Post-Marketing Pharmacovigilance And Medical Information Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Post-Marketing Pharmacovigilance And Medical Information Market 2026, By The Business Research Company
Drain Cleaning Equipment Market Report 2026
https://www.thebusinessresearchcompany.com/report/drain-cleaning-equipment-global-market-report
Urinary Drainage Bags Market Report 2026
https://www.thebusinessresearchcompany.com/report/urinary-drainage-bags-global-market-report
Prefabricated Buildings Market Report 2026
https://www.thebusinessresearchcompany.com/report/prefabricated-buildings-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at: marketing@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model