Rising adverse drug reactions are driving the Post-Inflammatory Hyperpigmentation Treatment Market 2026
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Market Size Expansion Is Predicted For The Post-Marketing Pharmacovigilance And Medical Information Market Between 2026 And 2030?
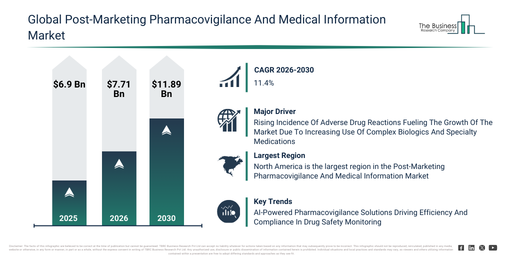
The post-marketing pharmacovigilance and medical information market has expanded rapidly in recent years. This market is expected to grow from $6.9 billion in 2025 to $7.71 billion in 2026, achieving a compound annual growth rate (CAGR) of 11.7%. The expansion observed in the past can be attributed to reasons such as limited post-marketing surveillance infrastructure, reliance on manual ADR reporting, increasing regulatory requirements for drug safety, a greater number of new drug approvals, and a lack of integrated hospital data systems.
The post-marketing pharmacovigilance and medical information market is projected for substantial expansion over the coming years. Its valuation is anticipated to reach $11.89 billion by 2030, demonstrating a robust compound annual growth rate (CAGR) of 11.4%. This anticipated growth during the forecast period is propelled by factors such as the integration of AI and ML algorithms for enhanced signal detection, the broader adoption of longitudinal patient data analysis, the expansion of disease registry-linked monitoring, the surge in digital app-based reporting, and increased investments in real-time pharmacovigilance technologies. Furthermore, significant trends expected over this period encompass a growing adoption of electronic health record (EHR) mining, an increasing deployment of real-time adverse drug reaction (ADR) monitoring systems, a rise in reporting categorized by risk population and therapeutic class, the broadening of cohort event monitoring for new drug users, and the seamless integration of digital and mobile platforms for pharmacovigilance reporting.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=27393&type=smp
What Major Drivers Are Influencing Demand In The Post-Marketing Pharmacovigilance And Medical Information Market?
The increasing occurrence of adverse drug reactions is anticipated to fuel the expansion of the post-marketing pharmacovigilance and medical information market in the future. These reactions are defined as detrimental or unforeseen effects arising from medication use at prescribed dosages for purposes such as prevention, diagnosis, or treatment. The frequency of adverse drug reactions is escalating because the elderly population increasingly needs to manage numerous chronic illnesses, which often involves taking multiple drugs concurrently, thereby increasing the risk of detrimental drug interactions and unforeseen side effects. Post-marketing pharmacovigilance and medical information services aid in handling adverse drug reactions through identifying, monitoring, and disseminating information about potential side effects once a drug is on the market, thus contributing to its continued safety and efficacy. For example, in January 2023, data from Navikenz, a US-based artificial intelligence-focused IT services company, indicated that adverse drug reactions in the US are projected to be responsible for 200,000 to 400,000 deaths annually, surpassing the combined fatalities from stroke and diabetes, with approximately 2.7 million annual cases leading to over 100,000 hospitalizations and more than 15,000 deaths. Consequently, the growing occurrence of adverse drug reactions is propelling the expansion of the post-marketing pharmacovigilance and medical information market.
Which Market Segments Are Examined In The Post-Marketing Pharmacovigilance And Medical Information Market Study?
The post-marketing pharmacovigilance and medical information market covered in this report is segmented –
1) By Type: Spontaneous Reporting, Intensified Adverse Drug Reaction (ADR) Reporting, Targeted Spontaneous Reporting, Cohort Event Monitoring, Electronic Health Record (EHR) Mining
2) By Publication Type: Books, Online Media, Journals
3) By End User: Hospitals, Research Organizations, Other End-Users
Subsegments:
1) By Spontaneous Reporting: Consumer Reporting, Healthcare Professional Reporting, Regulatory Authority Reporting, Pharmaceutical Company Reporting, Digital App-Based Reporting
2) By Intensified Adverse Drug Reaction (ADR) Reporting: Hospital-Based Surveillance, Program-Specific Monitoring, Disease-Specific Monitoring, Product-Specific Monitoring, Real-Time Monitoring Systems
3) By Targeted Spontaneous Reporting: Risk Population-Based Reporting, Therapeutic Class-Based Reporting, Adverse Drug Reaction Focused Reporting, Region-Specific Reporting, Healthcare Setting-Specific Reporting
4) By Cohort Event Monitoring: Prospective Cohort Monitoring, Retrospective Cohort Monitoring, Active Follow-Up Monitoring, New Drug User Monitoring, Disease Registry-Linked Monitoring
5) By Electronic Health Record (EHR) Mining: Natural Language Processing-Based Mining, Artificial Intelligence (AI) And Machine Learning Algorithms, Rule-Based Signal Detection, Longitudinal Patient Data Analysis, Integrated Hospital Data Systems
Which Trends Are Influencing The Post-Marketing Pharmacovigilance And Medical Information Market?
Companies operating within the post-marketing pharmacovigilance and medical information market are prioritizing the development of advanced solutions, such as AI-powered pharmacovigilance workflows, to bolster the accuracy of adverse event detection and enable real-time safety monitoring for improved patient outcomes. AI-powered pharmacovigilance workflows utilize artificial intelligence to automate the oversight of drug safety, thereby allowing for faster adverse event identification and enhanced regulatory compliance. For instance, in January 2024, PubHive Ltd., a UK-based cloud-based software-as-a-service (SaaS) company, introduced a centralized Summary of Product Characteristics (SmPC) management solution for life science companies and research organizations. This platform consolidates crucial drug safety information into a unified, easily accessible repository, automates literature monitoring and regulatory reporting, strengthens team collaboration, and simplifies compliance processes through advanced AI capabilities. This empowers pharmacovigilance teams to function more efficiently and proactively, ensuring both patient safety and adherence to regulatory standards.
Who Are The Primary Competitors In The Post-Marketing Pharmacovigilance And Medical Information Market?
Major companies operating in the post-marketing pharmacovigilance and medical information market are Accenture plc, Sanofi S.A., Capgemini SE, Merck & Co. Inc., Cognizant Technology Solutions Corporation, IQVIA Holdings Inc., HCL Technologies Limited, ICON plc, Syneos Health Inc., Genpact Limited, Parexel International Corporation, Avalere Health LLC, Quanticate Limited, Inizio Consulting LLC
Read the full post-marketing pharmacovigilance and medical information market report here:
Which Region Shows The Strongest Potential For Future Growth In The Post-Marketing Pharmacovigilance And Medical Information Market?
North America was the largest region in the post-marketing pharmacovigilance and medical information market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the post-marketing pharmacovigilance and medical information market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Post-Marketing Pharmacovigilance And Medical Information Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=27393&type=smp
Browse Through More Reports Similar to the Global Post-Marketing Pharmacovigilance And Medical Information Market 2026, By The Business Research Company
Drain Cleaning Equipment Market Report 2026
https://www.thebusinessresearchcompany.com/report/drain-cleaning-equipment-global-market-report
Urinary Drainage Bags Market Report 2026
https://www.thebusinessresearchcompany.com/report/urinary-drainage-bags-global-market-report
Prefabricated Buildings Market Report 2026
https://www.thebusinessresearchcompany.com/report/prefabricated-buildings-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model