Key Factors Fueling the Growth of the Preclinical CRO Market in 2025: Surging Demand For Preclinical Trials Drives Growth In The Preclinical CRO Market
Claim your 30% discount on Global Market Reports with code ONLINE30. Limited time only.
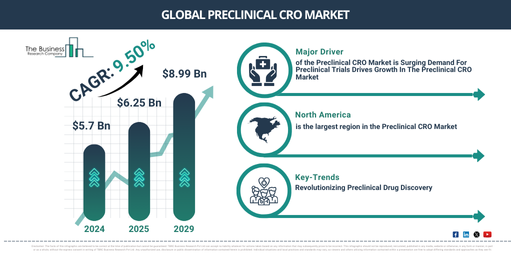
How Fast Is The Preclinical CRO Market Expected to Grow Between 2025 And 2029?
The size of the preclinical contract research organization (CRO) market has experienced substantial growth in previous years. It is projected to escalate from $5.7 billion in 2024 to $6.25 billion in 2025 with a compound annual growth rate (CAGR) of 9.7%. The boost witnessed in the historical era can be accredited to the increasing need for rapid drug development, strategies for cost containment, concentration on core competencies, compliance with regulatory demands, and the internationalization of clinical trials.
The size of the preclinical CRO market is anticipated to witness robust expansion in the coming years. By 2029, it is projected to reach a value of$8.99 billion” with a compound annual growth rate (CAGR) of 9.5%. This anticipated growth during the forecast period is due to the surge in personalized medicine, concentrated efforts on rare diseases and orphan drugs, increased outsourcing in the field of biopharmaceuticals, a strong focus on biomarker research and a heightened demand for real-world evidence generation. Future trends in this period are predicted to include the incorporation of artificial intelligence (AI), the enlargement of oncology research, progress in omics technologies, advanced in vitro and in vivo models, and collaborations for translational research.
Download a free sample to assess the report’s scope and structure:
https://www.thebusinessresearchcompany.com/sample.aspx?id=10303&type=smp
What are the Fundamental Drivers and Innovations Shaping the Preclinical CRO Market?
The rising necessity for preclinical trials is believed to spur the expansion of the preclinical CRO market in the future. Preclinical studies, which refer to any research conducted on a treatment or medicine prior to its testing on human subjects, often encompass new medical devices, prescription drugs, and diagnostics. The principal goals are to establish a safe starting dosage for initial human research and assess any potential toxicity of the product. Given their critical role in drug development, and the growing focus on the safety and efficacy of new drugs, the importance of preclinical trials is magnifying. For example, USA-based IQVIA reported in February 2024 that clinical development efficiency climbed in 2023, with the global launch of 69 novel active substances (NASs), six more than the previous year, including 24 first-in-class launches in the US. Consequently, the escalating need for preclinical trials is directing the upward trend of the preclinical CRO market.
How Is the Preclinical CRO Market Segmented?
The preclinical cromarket covered in this report is segmented –
1) By Service: Bioanalysis And DMPK Studies; Toxicology Testing; Compound Management; Chemistry; Safety Pharmacology; Others Services
2) By Type: Patient Derived Organoid (PDO) Model; Patient Derived Xenograft Model
3) By Animal Model: Small Animal Model; Large Animal Model
4) By Model System: In Vivo; In Vitro
5) By End User: Biopharmaceutical Companies; Government And Academic Institutes; Medical Device Companies; Other End-users
Subsegments:
1) By Bioanalysis And DMPK Studies: Pharmacokinetics Studies; Pharmacodynamics Studies; Drug Metabolism Studies
2) By Toxicology Testing: Acute Toxicity Testing; Chronic Toxicity Testing; Reproductive Toxicity Testing
3) By Compound Management: Sample Management; Inventory Management; Formulation Services
4) By Chemistry: Medicinal Chemistry; Analytical Chemistry; Synthetic Chemistry
5) By Safety Pharmacology: Cardiovascular Safety Studies; Central Nervous System Safety Studies; Respiratory Safety Studies
6) By Other Services: Regulatory Consulting; Project Management; Data Management Services
Request customized data on this market:
https://www.thebusinessresearchcompany.com/customise?id=10303&type=smp
Which Regions Are Driving the Next Phase of the Preclinical CRO Market Growth?
North America was the largest region in the preclinical CRO market in 2024. Asia-Pacific is expected to be the fastest-growing region in the global preclinical CRO market during the forecast period. The regions covered in the preclinical CRO market report are Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa
What Key Trends Are Shaping the Future of the Preclinical CRO Market?
Emerging technologies are becoming increasingly popular as a significant trend in the preclinical CRO sector. Many dominant players in this market are leveraging new technologies to maintain their market standings. For instance, in February 2023, BenchSci, an AI solutions firm for preclinical R&D based in Canada, introduced ASCEND, a comprehensive SaaS (software as a service) platform designed for preclinical drug discovery. The aim is to accelerate preclinical phase drug development pipelines by uncovering biological insights into the root causes of diseases. The ASCEND platform, provided by BenchSci, uses machine learning technology to source experimental data from reliable internal and public external resources. It utilizes curated ontology datasets to compare experimental outcomes. Consequently, the software can generate a map of the biological mechanisms behind various diseases based on existing evidence.
View the full report here:
https://www.thebusinessresearchcompany.com/report/preclinical-cro-global-market-report
How Is the Preclinical CRO Market Defined and What Are Its Core Parameters?
Preclinical CROs (contract research organizations) are support organizations that provide the knowledge in research and development needed to guide a drug candidate through animal testing and advance it to the clinical stage. A preclinical CRO supports new medical product producers in showing product safety and efficacy in living animals that the Food and Drug Administration (FDA) considers mirroring human anatomy most closely before entering clinical trials or being used for human care.
Purchase the full report and get a swift delivery:
https://www.thebusinessresearchcompany.com/purchaseoptions.aspx?id=10303
About The Business Research Company:
With over 15000+ reports from 27 industries covering 60+ geographies, The Business Research Company has built a reputation for offering comprehensive, data-rich research and insights. Armed with 1,500,000 datasets, the optimistic contribution of in-depth secondary research, and unique insights from industry leaders, you can get the information you need to stay ahead in the game.
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Europe +44 7882 955267
Asia & Others +44 7882 955267 & +91 8897263534
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model