Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market Forecast: How Big Will It Be by 2030?
Uncover key drivers, emerging technologies, and competitive movements shaping the programmed death (pd)-1 non-small cell lung cancer market from 2026–2035 with trusted insights from The Business Research Company
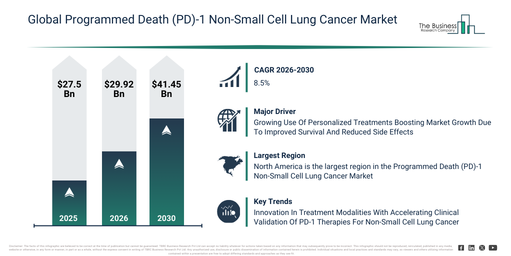
By how much is the Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market expected to grow between 2026 and 2030?
The market size for programmed death (pd)-1 non-small cell lung cancer has demonstrated strong growth in recent years. It is projected to expand from $27.5 billion in 2025 to $29.92 billion in 2026, at a compound annual growth rate (CAGR) of 8.8%. This historical growth can be attributed to various factors, including the limited number of pd-1 inhibitors approved for nsclc, the increasing incidence of lung cancer, growing physician awareness concerning immune checkpoint therapies, the reliance on chemotherapy as standard care, and the early clinical trial successes of pembrolizumab and nivolumab.
The programmed death (pd)-1 non-small cell lung cancer market size is projected for significant expansion in the next few years, forecast to reach $41.45 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 8.5%. This growth during the forecast period is attributed to the creation of novel combination therapies, the broader application of biomarker-driven personalized treatment, increasing adoption of mRNA vaccine-based therapies, growing investment in immuno-oncology research, and rising government and private funding for cancer immunotherapy. Major trends anticipated in this period include an increased uptake of pd-1 monotherapy for NSCLC, a rise in combination therapies utilizing chemotherapy, CTLA-4 inhibitors, and VEGF inhibitors, augmented investment in targeted therapy and mRNA vaccine research, the expansion of both first-line and second-line treatment options, and the integration of personalized medicine and biomarker testing into treatment planning.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25888&type=smp
Which Drivers Are Influencing Market Acceleration In The Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market?
The programmed death (PD)-1 non-small cell lung cancer market is anticipated to expand due to the growing acceptance of personalized treatments. These treatments involve medical strategies customized to an individual’s unique genetic makeup, way of life, and specific disease characteristics, aiming for more precise and impactful results. The increased demand for personalized treatments stems from the wider accessibility of genomic profiling and biomarker testing, as these technologies facilitate the identification of particular mutations and biological indicators, enabling healthcare professionals to choose therapies with a higher probability of success for individual patients. For programmed death (PD)-1 non-small cell lung cancer (NSCLC), personalized treatments are beneficial as they customize therapies according to a patient’s genetic profile, tumor features, and immune system reaction. As an example, the Personalized Medicine Coalition, a US-based non-profit organization, reported in February 2024 that the US Food and Drug Administration (FDA) sanctioned 16 new personalized treatments for rare diseases in 2023, an increase from 6 in 2022. Consequently, the expanding embrace of personalized treatments is a key factor boosting the programmed death (PD)-1 non-small cell lung cancer market.
What Segments Are Identified Within The Structure Of The Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market?
The programmed death (pd)-1 non-small cell lung cancer market covered in this report is segmented –
1) By Drug Type: Monotherapy, Combination Therapy
2) By Line of Therapy: First-line Treatment, Second-line And Beyond
3) By End User: Hospitals, Specialty Cancer Centers, Academic And Research Institutes, Other End Users
Subsegments:
1) By Monotherapy: Pembrolizumab (Keytruda), Nivolumab (Opdivo), Cemiplimab (Libtayo), Toripalimab, Tislelizumab, Camrelizumab, Dostarlimab, Other Monotherapy Drug Types
2) By Combination Therapy: Programmed Death (PD)-1 Inhibitor And Chemotherapy, Programmed Death (PD)-1 Inhibitor And Cytotoxic T-Lymphocyte Associated Protein (CTLA)-4 Inhibitor, Programmed Death (PD)-1 Inhibitor And Vascular Endothelial Growth Factor (VEGF) Inhibitor, Programmed Death (PD)-1 Inhibitor And Targeted Therapy, Programmed Death (PD)-1 Inhibitor And Radiation Therapy, Triple Combination Therapy, Programmed Death (PD)-1 Inhibitor And Messenger Ribonucleic Acid (mRNA) Vaccine, Other Combination Drug Types
What Upcoming Trends Are Likely To Define The Future Path Of The Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market?
Major companies in the programmed death (PD)-1 non-small cell lung cancer market are focusing on conducting clinical trials for innovative therapies, such as monotherapy, to improve patient outcomes. Monotherapy is a treatment strategy that employs a single drug or therapy to address a disease or condition. For example, in March 2025, Akeso, Inc., a China-based biopharmaceutical company, announced positive results from its Phase III HARMONi-2 trial, which evaluated Ivonescimab (AK112) in combination with chemotherapy for the first-line treatment of advanced non-small cell lung cancer (NSCLC). The study successfully met its primary endpoint by significantly extending progression-free survival compared to pembrolizumab, highlighting the therapeutic potential of the dual-targeted bispecific antibody.
Which Major Firms Are Strengthening Their Position In The Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market?
Major companies operating in the programmed death (pd)-1 non-small cell lung cancer market are F. Hoffmann-La Roche Ltd, Merck & Co. Inc., Sanofi S.A., Novartis AG, GlaxoSmithKline plc, Regeneron Pharmaceuticals Inc., Incyte Corporation, Ono Pharmaceutical Co. Ltd., BeiGene Ltd., BioNTech SE, Shanghai Junshi Biosciences Co. Ltd., Akeso Inc., Arcus Biosciences Inc., Shandong Boan Biotechnology Co. Ltd., CStone Pharmaceuticals, iTeos Therapeutics Inc., Summit Therapeutics Inc., Shanghai Henlius Biotech Inc.
Access The Complete Report For Deeper Market Insights:
Which Region Shows The Strongest Potential For Future Expansion In The Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market?
North America was the largest region in the programmed death (PD)-1 non-small cell lung cancer market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the programmed death (pd)-1 non-small cell lung cancer market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=25888&type=smp
Browse Through More Reports Similar to the Global Programmed Death (PD)-1 Non-Small Cell Lung Cancer Market 2026, By The Business Research Company
Pd 1 And Pdl1 Inhibitors Or Immune Checkpoint Inhibitors Market Report 2026
Non Small Cell Lung Cancer Nsclc Market Report 2026
Small Cell Lung Cancer Therapeutics Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model