Rare Disease Clinical Trials Market Outlook 2026–2035 Driven by Key Growth Factors and Industry Projections
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
What Is The Market Size Of The Rare Disease Clinical Trials Market In 2026 And What Value Is Projected For 2030?
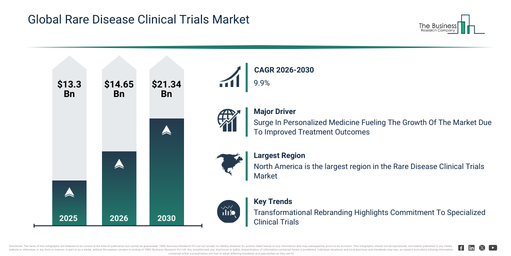
The rare disease clinical trials market size has experienced rapid growth in recent years. It is forecast to grow from $13.3 billion in 2025 to $14.65 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 10.1%. Historically, this expansion can be attributed to the broadening of orphan drug development programs, increased regulatory incentives supporting rare disease research, the rise of specialized clinical research organizations, enhanced collaboration between academia and industry, and more effective diagnostic identification of rare diseases.
The rare disease clinical trials market is projected to experience substantial expansion in the coming years. This market is anticipated to reach a value of $21.34 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 9.9%. This anticipated growth is driven by factors such as the wider uptake of virtual and hybrid trial models, the increasing application of AI for trial optimization, the broadening of global rare disease trial networks, the escalating need for quicker regulatory clearances, and increased funding for rare disease research and development. Key trends expected during this period involve the greater utilization of decentralized clinical trial frameworks, the increasing acceptance of adaptive trial designs, the deeper integration of digital tools for patient recruitment, the wider collection of real-world evidence, and an intensified focus on patient-centered trial protocols.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=28311&type=smp
Which Growth Drivers Are Contributing To The Progress Of The Rare Disease Clinical Trials Market?
The expansion of personalized medicine is anticipated to significantly advance the rare disease clinical trials market in the future. Personalized medicine involves customizing medical care based on each patient’s unique attributes, including their genetics, lifestyle choices, and environmental factors. The increasing prominence of personalized medicine is attributed to its capacity for improved treatment outcomes, as it tailors therapies to individual patient profiles for greater efficacy and safety. Rare disease clinical trials play a crucial role in advancing personalized medicine by generating patient-specific insights that guide the development of targeted therapies designed for each individual’s distinct genetic and molecular profile. For instance, in February 2024, the Personalized Medicine Coalition (PMC), a US-based nonprofit organization, reported that the Food and Drug Administration (FDA) noted personalized medicines accounted for 34% of approvals in 2022, a figure that climbed to 38% in 2023. Thus, the growth of personalized medicine is a key driver for the rare disease clinical trials market.
What Are The Key Segments Of The Rare Disease Clinical Trials Market?
The rare disease clinical trials market covered in this report is segmented –
1) By Phase: Phase I, Phase II, Phase III, Phase IV
2) By Therapeutic Area: Oncology, Cardiovascular Disorders, Neurological Disorders, Infectious Disease, Genetic Disorders, Autoimmune And Inflammation, Hematologic Disorders, Musculoskeletal Disorders, Other Therapeutic Areas
3) By Study Design: Interventional, Observational, Expanded Access
4) By End-User: Pharmaceutical Companies, Biotechnology Companies, Research Institutes, Other End-Users
Subsegments:
1) By Phase I: First In Human Trials, Dose Escalation Studies, Safety And Tolerability Studies
2) By Phase II: Proof Of Concept Studies, Dose Response Studies, Efficacy And Safety Studies
3) By Phase III: Randomized Controlled Trials, Comparative Effectiveness Studies, Large Scale Multicenter Trials
4) By Phase IV: Post Marketing Surveillance, Long Term Safety Studies, Real World Evidence Studies
How Are Trends Shaping The Direction Of The Rare Disease Clinical Trials Market?
Major companies within the rare disease clinical trials market are concentrating on driving innovations, including the implementation of a client-centric approach to provide personalized trial designs, boost patient engagement, and hasten the development of targeted therapies for rare conditions. A client-centric methodology entails prioritizing the requirements, goals, and preferences of biotech and pharmaceutical clients to deliver tailored and effective clinical trial solutions. For instance, in March 2025, Evestia Clinical Limited, a US-based pharmaceutical research company, was unveiled as the new brand identity of EMAS Pharma. This rebranding represents a strategic initiative aimed at accelerating the company’s global expansion. The company is committed to expediting the development of therapies for rare diseases by offering expert-driven, personalized clinical trial solutions specifically crafted for the unique challenges associated with these conditions. This particular emphasis is a core element of its brand identity and service offerings.
Who Are The Leading Companies Operating In The Rare Disease Clinical Trials Market?
Major companies operating in the rare disease clinical trials market are F. Hoffmann-La Roche AG, Pfizer Inc., Novartis AG, AstraZeneca plc, Takeda Pharmaceutical Company Limited, IQVIA Holdings Inc., Laboratory Corporation of America Holdings, ICON plc, Moderna Inc., Charles River Laboratories International Inc., Parexel International Corporation, Revvity Inc., Sarepta Therapeutics Inc., TFS HealthScience, Inventiva S.A., Tonix Pharmaceuticals Holding Corp., SpringWorks Therapeutics Inc., OrphAI Therapeutics Inc., BBCR Consulting LLC, Credevo Inc.
Read the full rare disease clinical trials market report here:
https://www.thebusinessresearchcompany.com/report/rare-disease-clinical-trials-global-market-report
What Are The Leading Geographic Regions In The Rare Disease Clinical Trials Market?
North America was the largest region in the rare disease clinical trials market in 2025. The regions covered in the rare disease clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Rare Disease Clinical Trials Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=28311&type=smp
Browse Through More Reports Similar to the Global Rare Disease Clinical Trials Market 2026, By The Business Research Company
Rare Inflammatory Disease Treatment Global Market Report
Veterinary Dermatology Drugs Global Market Report
https://www.thebusinessresearchcompany.com/report/veterinary-dermatology-drugs-global-market-report
Dermatophytosis Treatment Global Market Report
https://www.thebusinessresearchcompany.com/report/dermatophytosis-treatment-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model