The Ribonucleic Acid (RNA) Therapy Clinical Trials Market Is Witnessing Impact From Increase In Surgical Procedures Driving The Growth Of The Due To Rising Prevalence Of Chronic Diseases
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
What Are The Market Size Estimates For The Ribonucleic Acid (RNA) Therapy Clinical Trials Market In 2026 And 2030?
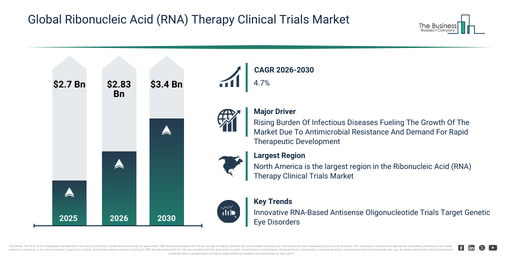
The market size for ribonucleic acid (rna) therapy clinical trials has demonstrated consistent growth in recent years. It is anticipated to expand from $2.7 billion in 2025 to $2.83 billion by 2026, progressing at a compound annual growth rate (CAGR) of 4.8%. Throughout its historical period, this growth can be attributed to elements such as limited rna-based therapeutic options, the high cost associated with early-stage clinical trials, difficulties in obtaining regulatory approval, a general lack of awareness regarding rna therapies, and a prevailing reliance on traditional therapeutic modalities.
The ribonucleic acid (rna) therapy clinical trials market size is anticipated to experience consistent expansion over the upcoming years. This market is projected to reach $3.4 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 4.7%. The expansion during this forecast period is primarily driven by improvements in RNA delivery technologies, increased investment in research for rare diseases and cancer, enhanced partnerships between biotechnology and pharmaceutical firms, the growing use of personalized medicine strategies, and the broadening of international clinical trial infrastructures. Key trends anticipated for the forecast timeframe encompass a rise in RNA therapy clinical trials across various treatment domains, an intensified focus on RNA therapeutics for rare diseases and cancer, the growth of mRNA vaccine and self-amplifying RNA therapy trials, the integration of CRISPR and RNA editing-based therapy trials, and the incorporation of sophisticated delivery platforms alongside oligonucleotide conjugates.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=25896&type=smp
What Leading Drivers Are Supporting The Ribonucleic Acid (RNA) Therapy Clinical Trials Market Expansion?
The rising prevalence of infectious diseases is projected to drive the expansion of the ribonucleic acid (RNA) therapy clinical trials market in the future. These diseases represent conditions resulting from harmful microorganisms, including bacteria, viruses, fungi, or parasites, which enter the body, reproduce, and negatively impact health. A primary contributor to the increase in infectious diseases is the growing volume of international travel, accelerating the swift transmission of pathogens across various geographical areas and communities. Ribonucleic acid (RNA) therapy clinical trials assist in confronting infectious diseases through facilitating the creation and assessment of specific treatments capable of adapting rapidly to newly identified pathogens. As an illustration, in April 2025, a report from the Minnesota Department of Health, a US-based state agency, indicated that in 2024, the percentage of HIV cases among males aged 35 to 39 rose from 10% in 2023 to 17%. Consequently, the growing impact of infectious diseases is fueling the expansion of the ribonucleic acid (RNA) therapy clinical trials market.
How Are Segments Identified Within The Ribonucleic Acid (RNA) Therapy Clinical Trials Market Segment Framework?
The ribonucleic acid (rna) therapy clinical trials market covered in this report is segmented –
1) By Modality: Ribonucleic Acid (RNA) Interference, Antisense Therapy, Messenger Ribonucleic Acid (RNA), Oligonucleotide, Non-Antisense, Non-Ribonucleic Acid Interference (RNAi)
2) By Clinical Trials Phase: Phase I, Phase II, Phase III, Phase IV
3) By Therapeutic Areas: Rare Diseases, Anti-Infective, Anticancer, Neurological, Alimentary Or Metabolic, Musculoskeletal, Cardiovascular Respiratory, Sensory, Other Therapeutic Areas
Subsegments:
1) By Ribonucleic Acid (RNA) Interference: Small Interfering RNA (siRNA) Therapy Trials, MicroRNA (miRNA)-Based Therapy Trials, Short Hairpin RNA (shRNA) Therapy Trials, Dicer Substrate RNA Therapy Trials
2) By Antisense Therapy: Gapmer Antisense Oligonucleotide Trials, Steric Blocking Antisense Trials, Splice-Switching Antisense Oligonucleotide Trials, Antisense Oligonucleotide-Conjugated Delivery Trials
3) By Messenger Ribonucleic Acid (RNA): mRNA-Based Vaccines Trials, Self-Amplifying mRNA (saRNA) Therapy Trials, Non-Replicating mRNA Therapy Trials, Circular mRNA Therapy Trials
4) By Oligonucleotide: Phosphorothioate Oligonucleotide Trials, Peptide Nucleic Acid (PNA) Trials, Locked Nucleic Acid (LNA) Trials, Morpholino Oligonucleotide Trials
5) By Non-Antisense: RNA Aptamer Therapy Trials, Guide RNA (gRNA)-Mediated CRISPR Trials, RNA Editing Therapy Trials, RNA Scaffolding Or Regulatory RNA Therapy Trials
6) By Non-Ribonucleic Acid Interference (RNAi): Ribozymes Therapy Trials, RNA Decoys Or Sponge RNA Trials, Long Non-Coding RNA (lncRNA) Therapy Trials, Small Activating RNA (saRNA) Therapy Trials
What Trends Are Shaping The Future Of The Ribonucleic Acid (RNA) Therapy Clinical Trials Market?
Leading entities within the ribonucleic acid (RNA) therapy clinical trials market are concentrating on creating novel solutions, including RNA-based antisense oligonucleotide therapy clinical trials, designed to pinpoint particular genetic sequences, regulate gene expression, and provide highly accurate treatments for both genetic and rare conditions. Such a clinical trial involves research into employing short synthetic RNA strands to attach to specific messenger RNA targets, with the goal of altering gene expression and addressing genetic ailments. As an illustration, during December 2024, Sepul Bio, a biotechnology firm based in France, commenced the LUNA Phase 2b clinical trial to assess ultevursen, an RNA-based antisense oligonucleotide therapy. This therapy is aimed at mutations in exon 13 of the USH2A gene, which are responsible for retinitis pigmentosa (RP) and Usher syndrome type 2a. This particular study, structured as a two-year, double-masked, randomized, sham-controlled investigation, plans to recruit 81 adults and children who are eight years old or older, across numerous international locations. The treatment is designed for individuals suffering from RP or non-syndromic RP resulting from exon 13 mutations in USH2A, a condition identifiable through genetic testing. The LUNA trial proceeds after encouraging outcomes from previous Phase 1/2 studies, in which ultevursen demonstrated improvements in visual acuity, retinal sensitivity, and retinal structure.
Who Are The Companies Competing Within The Ribonucleic Acid (RNA) Therapy Clinical Trials Market?
Major companies operating in the ribonucleic acid (rna) therapy clinical trials market are Pfizer Inc., AstraZeneca plc, Novartis AG, GlaxoSmithKline plc, Moderna Inc., BioNTech SE, Biogen Inc., Daiichi Sankyo Company Limited, Alnylam Pharmaceuticals Inc., Sarepta Therapeutics Inc., Ionis Pharmaceuticals Inc., Arrowhead Pharmaceuticals Inc., Arcturus Therapeutics Holdings Inc., Cartesian Therapeutics Inc., CureVac N.V., Orna Therapeutics Inc., Silence Therapeutics plc, ETHRIS GmbH, ProQR Therapeutics N.V., Lexeo Therapeutics Inc., Avidity Biosciences Inc., Aro Biotherapeutics Company, Wave Life Sciences Ltd.
Get The Full Ribonucleic Acid (RNA) Therapy Clinical Trials Market Report:
Which Region Holds The Highest Market Share In The Ribonucleic Acid (RNA) Therapy Clinical Trials Market?
North America was the largest region in the ribonucleic acid (RNA) therapy clinical trials market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the ribonucleic acid (rna) therapy clinical trials market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Ribonucleic Acid (RNA) Therapy Clinical Trials Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Ribonucleic Acid (RNA) Therapy Clinical Trials Market 2026, By The Business Research Company
Ribose Nucleic Acid Rna Based Therapeutics Market Report 2026
Nucleic Acid Based Gene Therapy Market Report 2026
Ribonucleic Acid Rna Sequencing Market Report 2026
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at: marketing@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model