Specimen Validity Testing Global Forecast: Market to Scale Up to $5.95 Billion by 2030 at 7.8% CAGR
Uncover key drivers, emerging technologies, and competitive movements shaping the specimen validity testing market from 2026–2035 with trusted insights from The Business Research Company
What is the estimated market size of the Specimen Validity Testing Market by the end of 2030?
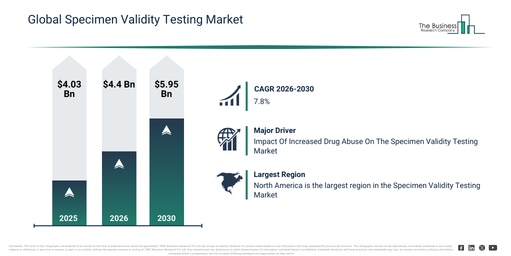
The specimen validity testing market has experienced significant growth in recent years. Its size is anticipated to expand from $4.03 billion in 2025 to $4.4 billion in 2026, achieving a compound annual growth rate (CAGR) of 9.3%. The market’s past development can be attributed to several factors including an increase in workplace drug testing, regulatory requirements for urine testing, a rise in cases of substance abuse, the demand for forensic testing, and the expansion of clinical laboratories.
The specimen validity testing market size is anticipated to experience substantial growth in the upcoming years. It is predicted to expand to $5.95 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 7.8%. This projected increase during the forecast period stems from the adoption of oral fluid testing, technological improvements in assay sensitivity, a rise in probation and legal testing requirements, the broadening of point-of-care solutions, and penetration into emerging markets. Significant trends expected during this period include an increase in adulterant detection panels, the automation of validity testing workflows, integration with laboratory information systems, the introduction of rapid testing kits, and an expansion of workplace drug screening initiatives.
Access Your Free Sample Report for In-Depth Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=15673&type=smp
What Drivers Are Shaping Future Opportunities In The Specimen Validity Testing Market?
The rise in drug abuse is projected to fuel the expansion of the Specimen Validity Testing Market in the foreseeable future. Drug abuse pertains to the habitual, excessive, or improper utilization of legal or illicit substances for non-medical reasons. The increase in drug abuse stems from various interconnected elements, including opioid addiction, escalating mental health problems, insufficient access to treatment and support, and societal portrayals. Specimen validity testing (SVT) techniques are designed to detect the presence of contaminants and compounds by analyzing multiple parameters like pH, specific gravity, creatinine content, and temperature. For instance, in October 2024, data from the Office for National Statistics, a UK-based government agency, indicated that England and Wales registered 5,448 drug poisoning-related deaths in 2023, representing an 11.0% increase from the 4,907 deaths recorded in 2022. Consequently, the growing prevalence of drug abuse is anticipated to drive the growth of the Specimen Validity Testing Market.
Which Segments Are Gaining Traction In The Specimen Validity Testing Market?
The specimen validity testing market covered in this report is segmented –
1) By Product And Service: Products, Service
2) By Testing Type: Laboratory Testing, Rapid Or Point-Of-Care Testing
3) By End-User: Drug Rehabilitation Centers, Drug Screening Laboratories, Pain Management Centers, Criminal Justice And Law Enforcement Agencies, Other End-Users
Subsegments:
1) By Products: Reagents And Assay Kits, Controls And Calibrators, Specimen Collection Devices, Analyzers And Instruments
2) By Service: Laboratory Testing Services, Point-Of-Care Testing Services
Who Are The Primary Competitors In The Global Specimen Validity Testing Market?
Major companies operating in the specimen validity testing market are Thermo Fisher Scientific Inc., Abbott Laboratories, Danaher Corporation, Siemens Healthineers , Medline Industries Inc., Becton, Dickinson and Company, Laboratory Corporation, Quest Diagnostics Incorporated, Alere Inc., Clinical Reference Laboratory Inc., Precision Diagnostics, ACM Global Laboratories, Millennium Health, Omega Laboratories Inc., Dominion Diagnostics, CannAmm, Sciteck Inc., Starplex Scientific Inc., Vision Diagnostics Inc., Premier Biotech Inc., Alfa Scientific Designs, Express Diagnostics, American Bio Medica Corporation, Titan Biotech Ltd., SureHire, Miraclean Technology Co. Ltd
Access The Complete Report For Deeper Market Insights:
https://www.thebusinessresearchcompany.com/report/specimen-validity-testing-global-market-report
Which Global Regions Are Shaping The Competitive Landscape Of The Specimen Validity Testing Market?
North America was the largest region in the specimen validity testing market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the specimen validity testing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request Your Customized Specimen Validity Testing Market Research Report for Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=15673&type=smp
Browse Through More Reports Similar to the Global Specimen Validity Testing Market 2026, By The Business Research Company
Virology Specimen Collection Market Report 2026
https://www.thebusinessresearchcompany.com/report/virology-specimen-collection-global-market-report
Laboratory Proficiency Testing Market Report 2026
Dna Testing Market Report 2026
https://www.thebusinessresearchcompany.com/report/dna-testing-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model