Viral and Non-Viral Vector Manufacturing Market Growth Driven by Increasing Clinical Trial Activity
The Business Research Company’s 2026 market reports feature enhanced tools like market attractiveness analysis, TAM assessment, and company scoring matrices, along with interactive dashboards, deeper supply chain insights, and startup coverage strengthening the depth, usability, and strategic value of insights.
How Will The Market Value Of The Viral And Non-Viral Vector Manufacturing Market Progress Between 2026 And 2030?
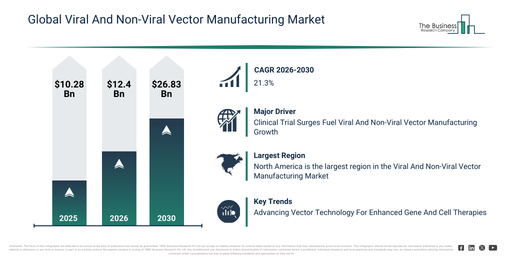
The viral and non-viral vector manufacturing market has experienced rapid expansion in recent years. This market is projected to expand from $10.28 billion in 2025 to $12.4 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 20.7%. Historically, this growth was driven by several factors, including advancements in gene therapy research, increased activity in clinical trials, the expansion of biopharmaceutical production facilities, a surging need for viral vectors in oncology, and the presence of specialized manufacturing expertise.
The viral and non-viral vector manufacturing market size is projected to experience substantial growth in the coming years. It is anticipated to expand to $26.83 billion by 2030, achieving a compound annual growth rate (CAGR) of 21.3%. The growth observed throughout the forecast period can be attributed to the rising commercialization of gene and cell therapies, increased investments in vaccine development, the broadening of CDMO partnerships, a heightened focus on scalable and flexible manufacturing approaches, and an increase in regulatory approvals for advanced therapies. Key trends during this period include the intensifying scale-up of viral vector production capacity, a growing demand for non-viral delivery systems, the wider adoption of modular manufacturing platforms, an expansion of contract vector manufacturing services, and an enhanced emphasis on process standardization and quality control.
Access Your Free Sample Report For In-Depth Market Analysis:
https://www.thebusinessresearchcompany.com/sample.aspx?id=12570&type=smp
Which Drivers Are Supporting The Viral And Non-Viral Vector Manufacturing Market Growth?
An anticipated increase in clinical trials is projected to fuel the expansion of the viral and non-viral vector manufacturing markets in the future. These trials are research endeavors involving human subjects to assess the safety, effectiveness, and possible adverse effects of medical interventions, treatments, or drugs. The production of viral and non-viral vectors is crucial for gene therapy clinical trials, as it supplies the essential components for delivering therapeutic genes to patients and guarantees the safety, quality, and scalability of these vectors across all trial stages. For example, data from ClinicalTrials.gov, a US-based online platform managed by the US National Library of Medicine, indicated that by December 2023, for the year 2022, a total of 437,515 clinical trials had been registered, with 56,561 successfully concluded. By December 20, 2023, the registration and completion figures had both increased, reaching 476,626 registered clinical trials and 61,504 completed trials. Consequently, the increasing volume of clinical trials is a key driver for the expansion of the viral and non-viral vector manufacturing markets.
What Are The Key Segment Divisions In The Viral And Non-Viral Vector Manufacturing Market Segment Structure?
The viral and non-viral vector manufacturing market covered in this report is segmented –
1) By Vector Type: Viral Vector, Non-Viral Vector
2) By Disease: Cancer, Genetic Disease, Infectious Disease, Cardiovascular Disease, Other Diseases
3) By Application: Gene Therapy, Vaccinology, Cell Therapy, Other Applications
Subsegments:
1) By Viral Vector: Adenoviral Vectors, Lentiviral Vectors, AAV (Adeno-Associated Virus) Vectors, Retroviral Vectors, Other Viral Vectors
2) By Non-Viral Vector: Plasmid DNA, Lipid-Based Vectors, Nanoparticles, Electroporation-Based Vectors, Other Non-Viral Vectors
Which Trends Are Expected To Impact The Viral And Non-Viral Vector Manufacturing Market?
Leading companies in the viral and non-viral vector manufacturing market are primarily focused on forming alliances to enhance their profitability. These collaborative partnerships in viral and non-viral vector manufacturing bring together biopharmaceutical companies, CDMOs, research institutions, and technology providers, all working to improve the efficiency, safety, and scalability of non-viral vector development and production. For instance, in September 2023, NecstGen, a Netherland-based CDMO that offers extensive support for Cell and Gene Therapy developers, partnered with ProteoNic BioSciences, a Netherland-based technology provider specializing in complex therapeutic protein production, to launch ProteoNic’s 2G UNic premium vector technology. The successful deployment of this technology has improved the development of lentiviral (LV) vectors, increasing functional viral particle titers and directly boosting process efficiency, which subsequently enhances the cost-effectiveness and accessibility of various therapies. This adaptable technology is applicable to several areas, including viral vector production for gene therapy, mRNA therapeutics, and transient protein production. By advancing AAV and LV viral vector manufacturing technology for both transient systems and stable producer cell lines, this development has the capacity to benefit viral vector manufacturing for gene therapies and non-vector processes, providing potential advantages across a wide range of applications.
Which Organizations Are Engaged In The Viral And Non-Viral Vector Manufacturing Market?
Major companies operating in the viral and non-viral vector manufacturing market are Novartis AG, Thermo Fisher Scientific Inc., Merck KGaA, FUJIFILM Holdings Corporation, Regeneron Pharmaceuticals Inc., Astellas Pharma Inc., Lonza Group AG, WuXi AppTec Co Ltd., Catalent Inc., Sartorius AG, Charles River Laboratories Inc., Sarepta Therapeutics Inc., GenScript ProBio Co. Ltd., TAKARA BIO Inc., Oxford BioMedica plc, Beam Therapeutics Inc., Intellia Therapeutics Inc., MaxCyte Inc., Voyager Therapeutics Inc., Genethon, MeiraGTx Holdings plc, Evox Therapeutics Ltd., Entos Pharmaceuticals Inc., LogicBio Therapeutics Inc., bluebird bio Inc., 4D Molecular Therapeutics Inc., Precision NanoSystems Inc., GenSight Biologics S.A., Abeona Therapeutics Inc., Generation Bio Co.
Get The Full Viral And Non-Viral Vector Manufacturing Market Report:
Which Region Dominates The Viral And Non-Viral Vector Manufacturing Market By Market Share?
North America was the largest region in the viral and non-viral vector manufacturing market in 2025. The regions covered in the viral and non-viral vector manufacturing market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Customized Viral And Non-Viral Vector Manufacturing Market Report For Competitive Insights:
Browse Through More Reports Similar to the Global Viral And Non-Viral Vector Manufacturing Market 2026, By The Business Research Company
Viral Vector Manufacturing Market Report 2026
https://www.thebusinessresearchcompany.com/report/viral-vector-manufacturing-global-market-report
Oral Expectorant Market Report
https://www.thebusinessresearchcompany.com/report/oral-expectorant-global-market-report
Viral Inactivation Market Report
https://www.thebusinessresearchcompany.com/report/viral-inactivation-global-market-report
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model