Technological innovation is advancing safety in the Viral Clearance Market 2026
The Business Research Company’s 2026 market reports include new capabilities such as market attractiveness scoring and analysis, total addressable market analysis, company scoring matrix, interactive excel data dashboard, improved supply chain analysis, upcoming startups in the market, and overview of key products, aimed at improving the depth, usability, and strategic value of the insights delivered.
How Will The Market Size Of The Viral Clearance Market Evolve From 2026 To 2030?
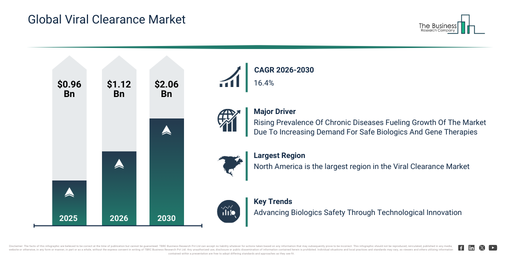
The viral clearance market has experienced significant growth in recent years. It is anticipated to increase from $0.96 billion in 2025 to $1.12 billion in 2026, demonstrating a compound annual growth rate (CAGR) of 16.7%. Historically, this expansion has been influenced by factors such as limited viral clearance technologies, increasing concerns about viral contamination in biologics, the ongoing reliance on conventional filtration methods, growing regulatory scrutiny, and the broadening of pharmaceutical and biotech manufacturing pipelines.
The viral clearance market is projected to experience substantial expansion over the upcoming years. This market is anticipated to reach a valuation of $2.06 billion by 2030, demonstrating a compound annual growth rate (CAGR) of 16.4%. Factors contributing to this growth during the forecast period include the advancement of next-generation viral inactivation techniques, the increasing use of nanofiltration and membrane chromatography, the expansion of biologics and gene therapy manufacturing, the incorporation of automation into viral clearance processes, and augmented investment in the development of viral detection assays. Key trends expected during this period encompass the growing uptake of sophisticated viral detection and inactivation technologies, a heightened demand for secure and virus-free vaccines and therapeutics, the broadening scope of cellular and gene therapy products that necessitate viral clearance, the incorporation of high-efficiency filtration and nanofiltration technologies, and an increasing reliance on contract research organizations for viral clearance services.
Download the Free Sample Report to Explore Key Market Insights:
https://www.thebusinessresearchcompany.com/sample.aspx?id=27444&type=smp
What Key Drivers Are Influencing The Growth Of The Viral Clearance Market?
The increasing incidence of chronic illnesses is anticipated to drive expansion in the viral clearance market moving ahead. These conditions are long-term health issues that evolve gradually, persisting for extended periods and often necessitating ongoing treatment or management. Worldwide, the occurrence of chronic diseases is on the rise, mainly fueled by an aging global population. Enhanced healthcare and improved living standards contribute to longer lifespans, which in turn leads to a greater incidence of age-associated conditions like cardiovascular diseases, diabetes, arthritis, and cancer. Viral clearance plays a vital role in the creation of biologics and gene therapies that address chronic diseases, guaranteeing both their safety and efficacy. As an illustration, in June 2023, the Institute for Health Metrics and Evaluation, a US-based public health research institute, disclosed that over half a billion people worldwide were afflicted with diabetes. This figure is projected to exceed double, reaching 1.3 billion people by 2050. Consequently, the growing occurrence of chronic diseases is propelling the expansion of the viral clearance market.
Which Segment Areas Are Covered In The Viral Clearance Market Analysis?
The viral clearance market covered in this report is segmented –
1) By Method: Viral Clearance Method, Viral Removal Method, Viral Inactivation Method, Viral Detection Method
2) By Application: Vaccines And Therapeutics, Blood And Blood Products, Cellular And Gene Therapy Products, Tissue And Tissue Products, Other Applications
3) By End-User: Pharmaceutical And Biotechnology Companies, Contract Research Organizations, Academic Research Institutes, Other End Users
Subsegments:
1) By Viral Clearance Method: Downstream Processing, Filtration, Chromatography, Precipitation, Nanofiltration
2) By Viral Removal Method: Membrane Chromatography, Depth Filtration, Nanofiltration, Ultrafiltration, Protein A Affinity Chromatography
3) By Viral Inactivation Method: Solvent Detergent Method, Low pH Treatment, Heat Treatment (Pasteurization), UV-C Irradiation, Caprylate Treatment
4) By Viral Detection Method: PCR (Polymerase Chain Reaction), ELISA (Enzyme-Linked Immunosorbent Assay), Nucleic Acid Testing (NAT), Cell-Based Assays, Transmission Electron Microscopy (TEM)
What Key Developments And Trends Are Impacting The Viral Clearance Market?
Prominent companies within the viral clearance market are concentrating on creating cutting-edge products, such as next-generation virus removal filters, to bolster viral safety and enhance biopharmaceutical manufacturing efficiency. These next-generation virus removal filters are sophisticated filtration devices engineered to effectively eliminate viruses from biopharmaceutical products, thereby guaranteeing product safety, accelerating filtration speeds, and supporting increased productivity in drug manufacturing. As an illustration, in October 2024, Asahi Kasei Medical Co., Ltd., a Japan-based medical and bioprocess solutions provider, introduced its next-generation Planova FG1 virus-removal filter. This filter boasts approximately seven-times greater flux than Planova BioEX, which helps to reduce filtration times, lower the risk of virus breakthrough, and sustain robust protein and virus-removal performance without the need for a prefilter. The FG1 is compatible with standard CIP/SIP processes and has commenced mass production in smaller sizes, with larger formats slated for future release to facilitate scale-up, allowing biomanufacturers to achieve higher throughput and faster cycle times.
Which Global Companies Are Actively Competing In The Viral Clearance Market?
Major companies operating in the viral clearance market are Merck Group, Thermo Fisher Scientific Inc., Asahi Kasei Corporation, SGS SA, Eurofins Scientific Inc., Lonza Group AG, WuXi AppTec, Catalent Biologics, Charles River Laboratories, Nelson Labs, Pall Corporation, Sartorius AG, Creative Biolabs, Texcell SA, Vironova AB.
Read the full viral clearance market report here:
https://www.thebusinessresearchcompany.com/report/viral-clearance-global-market-report
Which Region Is Expected To Register The Fastest Growth In The Viral Clearance Market?
North America was the largest region in the viral clearance market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the viral clearance market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
Request A Tailored Viral Clearance Market Research Report For Competitive Advantage:
https://www.thebusinessresearchcompany.com/customise?id=27444&type=smp
Browse Through More Reports Similar to the Global Viral Clearance Market 2026, By The Business Research Company
Wood Manufacturing Market Report 2026
https://www.thebusinessresearchcompany.com/report/wood-manufacturing-global-market-report
Wood Products Market Report 2026
https://www.thebusinessresearchcompany.com/report/wood-products-global-market-report
Wood Processing Market 2026
https://www.thebusinessresearchcompany.com/report/wood-processing-market
Get in touch with us:
The Business Research Company: https://www.thebusinessresearchcompany.com/
Americas +1 310-496-7795
Asia +44 7882 955267 & +91 8897263534
Europe +44 7882 955267
Email us at info@tbrc.info
Follow us on:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model